FDA Votes on Molunpiravir
post by Zvi · 2021-12-01T15:00:00.482Z · LW · GW · 3 commentsContents
High Level Summary Key Facts The Actual Decision Takeaways None 3 comments
While Paxlovid Remains Illegal and is expected to remain illegal for at least several weeks, the FDA did manage to finally meet to discuss whether or not to legalize the other Covid-19 treatment pill, Merck’s Molunpiravir. While later data reduced effectiveness estimates from 50% to 30%, that’s still much better than 0% and it uses a unique mechanism that can probably be profitably combined with other treatments, so one might naively think that after sufficient stalling for appearances this would be easy.
One would be wrong. The vote was 13-10, was restricted to those at high risk, and could easily have failed outright.
I may want to later refer back to this, so it’s splitting off into its own post.
High Level Summary
Usually we get live-blogs from Helen Branswell and Mattew Herper. They held off and only issued a summary post later on this time, perhaps because the meeting was too painful. Luckily, my commenter Weekend Editor is excellent at summarizing such meetings, so I’ll quote their summary in full, link goes to their full post. After the summary, I’ll note the salient other details in the full post.
Today the FDA’s AMBAC meeting voted to recommend molnupiravir get an emergency use authorization. But just barely: 13 yes, 10 no, 0 abstentions. And with a lot of caveats among the yes votes.
Some issues:
(a) The efficacy vs hospitalization was “wobbly”: the interim report had 48.3% (CL: 20.5% – 66.5%) efficacy, but when they added the rest of the data it was only 30.4% (CL: 1.0% – 51.1%). People thought this might mean there were responder/nonresponder populations, and nobody knew a biomarker to distinguish them.
This is an interesting mix of good and bad thinking. Yes, it’s possible that the 50% vs. 30% thing wasn’t random, but why is that an issue here?
If you have a drug with 30% efficacy, that’s good. If you have a drug that is 50% effective half the time and 10% effective the other half, depending on who it is used on, and you can’t tell who is who, then you still have a 30% effective drug.
The difference is that because of the way you are now stacking your coin flips, it feels like you are now giving a 10% effective drug to some people without knowing it? Or there’s the ‘problem’ that if you knew more you could differentiate between the two populations, so now you have an imperfect procedure and it wouldn’t be ‘ethical’ somehow to proceed, or you’re blameworthy when the drug doesn’t work in a particular case?
I’m glad this didn’t cause a veto for enough voters, but consider that if this wasn’t an emergency situation, it might have caused one, despite being good news since it opens up the possibility of doing better once we know more.
Then again, perhaps the argument is that experiments are illegal except for getting drugs approved, so if we approved the drug we’d never run the experiment? Which has a certain kind of dystopian logic to it, I suppose.
Of course, same as it ever was, we stopped the study when the early results looked so good, and now we’re saying the results are at most barely good enough…
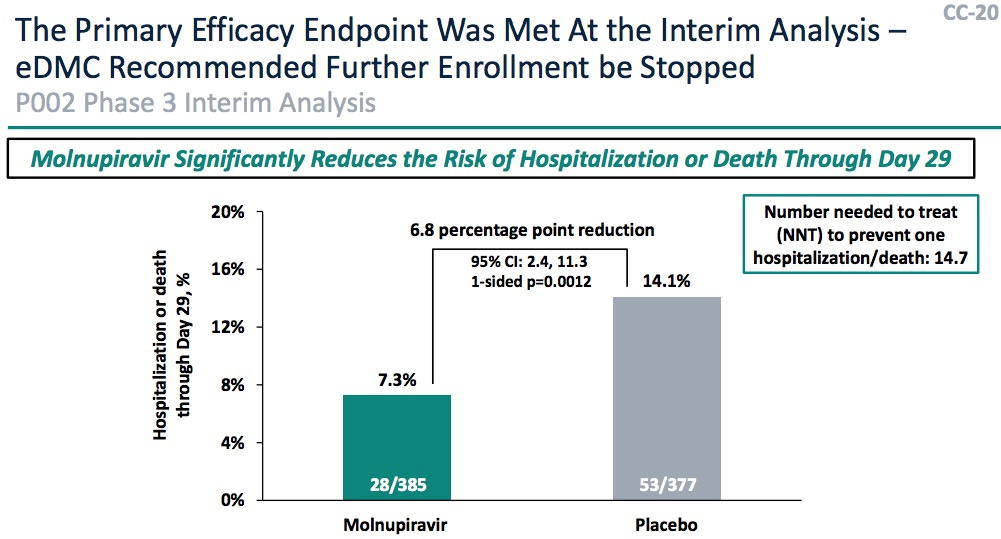
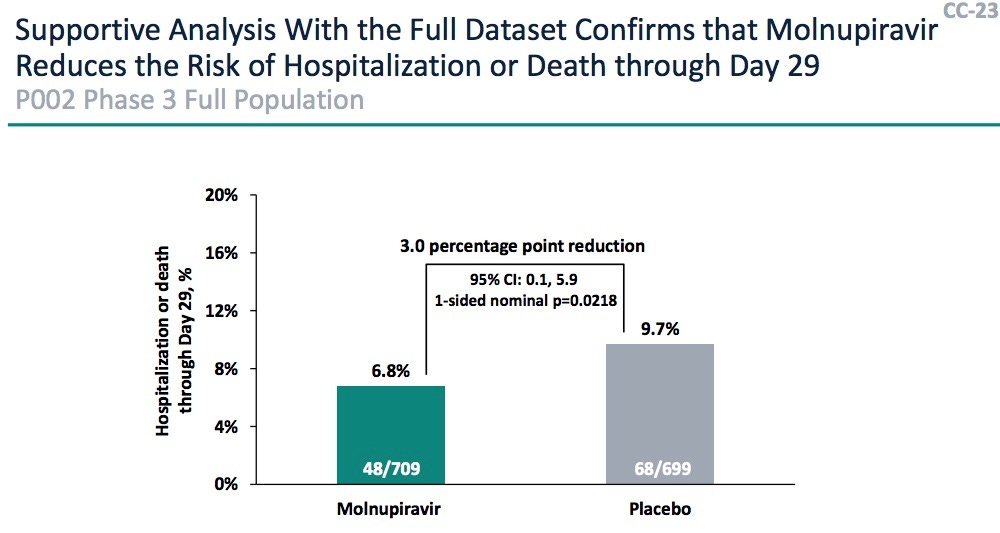
(b) While the mechanism and efficacy calculations from Merck were quite convincing, the FDA showed there were some issues with mutagenicity, particularly in the first trimester of pregnancy. (And I have to apologize: I got bored, and went grocery shopping at this point, stocking up in case the Omicronomicon gets loose. So I missed most of this, and am too tired to go back and listen. Maybe tomorrow.) Any woman will only get Molnupiravir after a negative pregnancy test.
Points for Omicronomicon, that’s great, and given the timing I hope it was all dry goods that will keep for a long time. In any case, a pregnancy test is quick and cheap, and damaged babies are considered quite bad, so I have no issue with this requirement, even though in practice it’s going to be painfully dumb a large percentage of the time. Given this is a treatment for those already sick, it would take a very large concern to make taking this a bad idea otherwise.
(c) One point in favor was that people thought the efficacy of monoclonal abs will fade with Omicron, so they want an alternative. OTOH, they also said if there’s “another oral medication” with higher efficacy with less side effects (think Paxlovid!), then the FDA should reconsider Molnupiravir. So Molnupiravir may get approved for like a month or so until Paxlovid blows it away? They so carefully didn’t mention Paxlovid that I wonder if there was some legal constraint.
This is of course completely crazy and illustrates how twisted their frameworks have become. Molnupiravir and Paxlovid are probably complements because they have different mechanisms, and the existence of a life saving medicine (that is currently illegal, thanks to you murderous madmen) is no reason to then make a different lifesaving medicine illegal because there’s a better option. If Paxlovid is legal and available and so is Molunpiravir, are they worried people who could have been given Paxlovid won’t be given Paxlovid?
Then there’s the crazy of the monoclonal abs argument and the ‘need’ for this new treatment as a justification. Once again, these treatments are complements, and also I can’t help but notice that a lot of people are dying of Covid-19 and we’re worried about a lack of hospital capacity and all that? It’s like the FDA thinks you only get one treatment (because who would dare use more than one without a Proper Scientific Study and a Standard of Care, or something) and therefore they have to ban all but the best treatment no matter the issues of cost and supply? The hell?
(d) Listening to the voting statements was almost painful: the AMDAC members were clearly conflicted.
So the recommended EUA only for high-risk individuals, mostly the unvaccinated or those who had suboptimal response to vaccination.
Pending FDA administrative action, Molnupiravir is EUA’d… sort of.
When they tried this insanity with boosters, limiting who can have access without legal liability to life saving medicine and thus allowing people to die, in order to satisfy arcane ‘ethical’ requirements, the states increasingly overruled the FDA. I very much hope that they do this again. But I fear that given the ‘mutagenicity’ issue doctors will rightfully fear lawsuits from people who claim nonsensical ‘mutations’ happened to them, and so won’t be able to give Molunpiravir to a lot of their patients, resulting in a bunch of people dying and hospitals filling up faster.
Seems like the most important action is to hurry along to the Paxlovid hearings, no?
Yes, still true, although they now have a distinct downside. They may lead to a lifesaving medicine, that is currently on track to be at least sort of legal, becoming illegal once more.
And it’s only on track to be legal rather than legal, with substantial doubt, because the FDA administer has to sign off, then the CDC advisory committee has to meet and then the CDC management has to approve. Given what we’ve seen so far, all of these steps are at risk.
However, it’s also important to note this concern, even if it didn’t make the comment summary and seems like it wasn’t considered too important:
There was also some concern that the induction of high mutation rates in the virus might, if it doesn’t go far enough, create a troublesome new variants.
This does seem like a real concern if it’s even slightly well-founded, and a good reason to consider not approving the drug. The downside of doing this could be very, very high. If this was the reason given for rejection, I might even accept it. Here’s what the Stat news summary had to report about that.
Panelists also worried about data showing that use of molnupiravir might, in theory, lead to new variants of the SARS-CoV-2 virus through its mechanism, which works by causing viruses to make mistakes in copying their genetic material.
“With all respect, I think it’s incumbent upon you to make some effort to make an estimate of what is the likelihood of escape mutants occurring as a result of your drug,” said James Hildreth, a panelist and the CEO of Meharry Medical College.
However, other panelists, including John Coffin, a Tufts molecular biologist, argued that the overall risk of such mutations due to the drug was small.
It seems appropriate here to have some amount of model error, and to actually do the calculation.
That’s the high level result.
Key Facts
There’s always a lot of good information at these meetings. What is it important to know?
Here’s the protocol:
The drug is administered 2ce per day for 5 days at a dose of 800mg (in the form of 4 capsules of 200mg each, so the dose can be titrated down, perhaps?).
Treatment must start within 5 days from symptoms. That’s empirically important, as shown here. The reduction in viral load over time is dramatic for intervention before 5 days past symptoms (left), but it is an order of magnitude weaker for intervention after 5 days of symptoms (right). That means we need to have a testing system which is widely available, cheap or free, and fast!
This also means that if you catch the problem quickly, we would probably see much better than 30% efficacy. This effect has to be continuous, there’s nothing special about five days in particular.
There were 10 subject deaths during the trial, but 9 were in the placebo arm. As a crude indication of safety, that’s pretty good. There were a lot of other safety studies, both in vitro, in animals, and in surveillance of the trial population. The trial seemed pretty safe, with adverse events in the placebo arm about the same as treatment. There is some worry about mutagenicity, particularly during organogenesis in pregnancy when messing with human RNA could be a bad idea.
The safety profile definitely more than passes my bar of ‘if this is unsafe then it’s nowhere near as unsafe as not using it so give it to me.’ If I get sick, I want treatment, and yet they are intending to make it illegal for me to get it, even outside issues of ‘delay.’
For a drug with 30% efficacy against hospitalization, that’s a rather good ratio. Sample size is small, but I suspect that the 30% is an underestimate.
There is no doubt this improvement is due to virus clearance, since they measured SARS-CoV2 RNA at baseline, day 3, and day 5. The reduction, as measured by log of mean difference and its 95% CL, is significant as shown here. It’s not just making people feel better, it’s doing so by a mechanism that makes sense and is related to the disease process.
It works. Weekend Editor notes this graph.
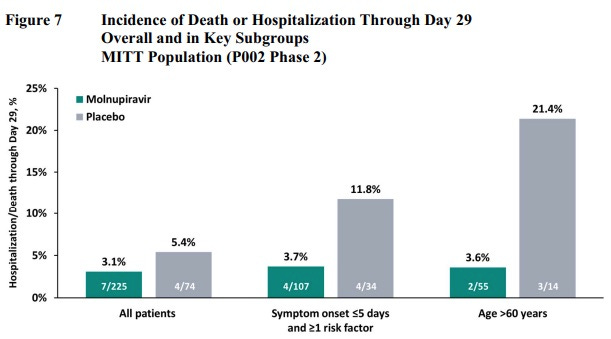
Weekend Editor suggests Molunpiravir might reduce everyone’s risk down to a similar baseline, based on this graph, which is interesting. Then again, have we considered that these sample sizes are too low and that’s why everything’s so noisy ? Still it is suggestive, and it is suggestive that the effective reduction in hospitalization and death could be much higher than 30% in practice.
This is the de rigeur Kaplan-Meier plot. It shows hospitalizations versus time, for the treatment arm and control arm. There’s a log-rank statistic that shows this difference is significant, i.e., the spread between the 2 curves is real.
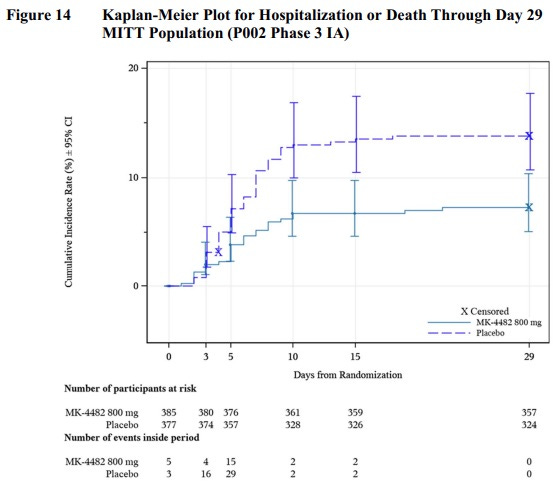
The logistical difficulties of getting the drug to people within 5 days of symptoms looked daunting to some AMDAC members.
If five days is daunting, how are we going to get Paxlovid to people within three? Also, seriously, five days is an insanely long amount of time, if it’s not enough then FIX IT.
The Actual Decision
It’s good to have a reference handy of how everyone voted, to compare with other votes in the future, or for other reasons.
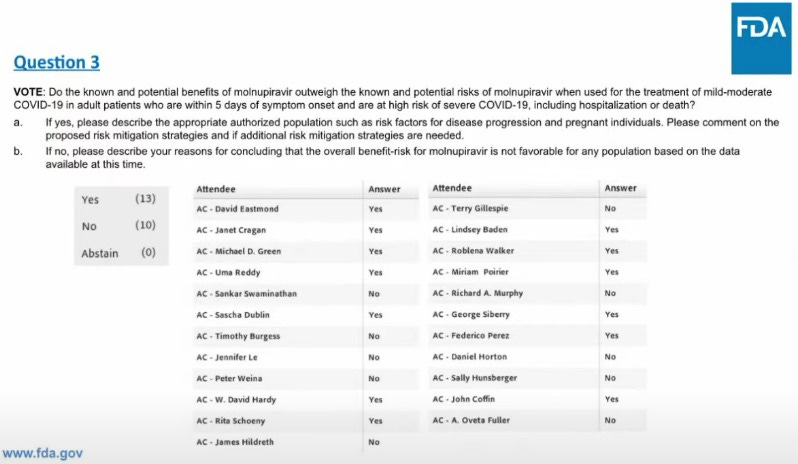
From the Stat news summary, some insight into what some people were thinking.
“I think we need to stop and acknowledge that the whole reason we’re having this discussion is because the efficacy of this product is not overwhelmingly good,” said W. David Hardy of Charles Drew University School of Medicine and Science during a discussion about the drug’s use during pregnancy. “And I think that makes all of us feel a bit uncomfortable about the fact whether this is an advance therapeutically because it’s an oral medication, not an intravenous medication.”
Then again that seems to directly contradict this summary viewpoint from the same article:
In the end, panelists narrowly voted that the benefits of having an oral Covid treatment to keep people out of the hospital outweighed their questions and concerns. But the FDA may write a far narrower authorization for the drug than observers would previously have expected.
This suggests that being an oral treatment was (correctly, I’d presume) considered an important advantage?
It also suggests that this decision was indeed very close and another similarly effective drug could easily have been rejected in this spot.
What is ‘overwhelmingly good?’ I’m guessing that if it were standard of care, and someone were suggesting not using it, the 30% would be enough to make this seem completely crazy and unacceptable. It’s all about framing.
It has to be ‘an advance’ because one does not simply use it in addition to other tools, and it’s a disadvantage that it is an oral medication in the context of whether it is ‘an advance’ in this other sense, even though in terms of usefulness it is a rather big advance, and the process is looking at a bunch of veto considerations rather than doing a cost-benefit analysis.
Takeaways
- When you stop a trial early, you sometimes don’t get enough data.
- This lack of data can then endanger approval. We have a concrete example.
- The vote was 13-10 and at best we’re likely to get a narrow authorization. If many of you want Molunpiravir, you won’t be able to get it, and it’s possible no one will be able to get it.
- The FDA is even more willing to deny us life saving medicine than we previously expected. We should worry more about this, not less.
- Pregnant women probably shouldn’t take Molunpiravir, and pregnancy tests will be required before the drug is given out.
- There is one other real potential worry about Molunpiravir, that it could create new variants. This does not seem to have been a major consideration, nor does a probability or cost-benefit assessment seem to have been done here, and the 13-10 vote was due mostly or entirely to other reasons.
- Chances seem good that this can be a >30% effective treatment in practice, among those who get it in time and are allowed to get it. I’d be far more surprised by substantially lower than 30% than by substantially higher.
- Paxlovid remains so illegal the FDA can’t even say its name in meetings.
3 comments
Comments sorted by top scores.
comment by Pattern · 2021-12-01T19:50:42.836Z · LW(p) · GW(p)
If you have a drug with 30% efficacy, that’s good. If you have a drug that is 50% effective half the time and 10% effective the other half, depending on who it is used on, and you can’t tell who is who, then you still have a 30% effective drug.
Worst case, you have a 10% effective drug. Do people want that? (Probably.)
Omicronomicon
?
Replies from: supposedlyfun↑ comment by supposedlyfun · 2021-12-01T23:20:45.270Z · LW(p) · GW(p)
Omicronomicon is a portmanteau of Omicron and Necronomicon, a book of evil magical power in the H.P. Lovecraft mythos.
comment by localdeity · 2021-12-01T23:44:44.142Z · LW(p) · GW(p)
Typo in the title (and 7 instances in text): "Molunpiravir" rather than "Molnupiravir".