Making Vaccine
post by johnswentworth · 2021-02-03T20:24:18.756Z · LW · GW · 249 commentsContents
The Process The Plan Motivations None 250 comments
Back in December, I asked [LW · GW] how hard it would be to make a vaccine for oneself. Several people pointed to radvac. It was a best-case scenario: an open-source vaccine design, made for self-experimenters, dead simple to make with readily-available materials, well-explained reasoning about the design, and with the name of one of the world’s more competent biologists (who I already knew of beforehand) stamped on the whitepaper. My girlfriend and I made a batch a week ago and took our first booster yesterday.
This post talks a bit about the process, a bit about our plan, and a bit about motivations. Bear in mind that we may have made mistakes - if something seems off, leave a comment.
The Process
All of the materials and equipment to make the vaccine cost us about $1000. We did not need any special licenses or anything like that. I do have a little wetlab experience from my undergrad days, but the skills required were pretty minimal.
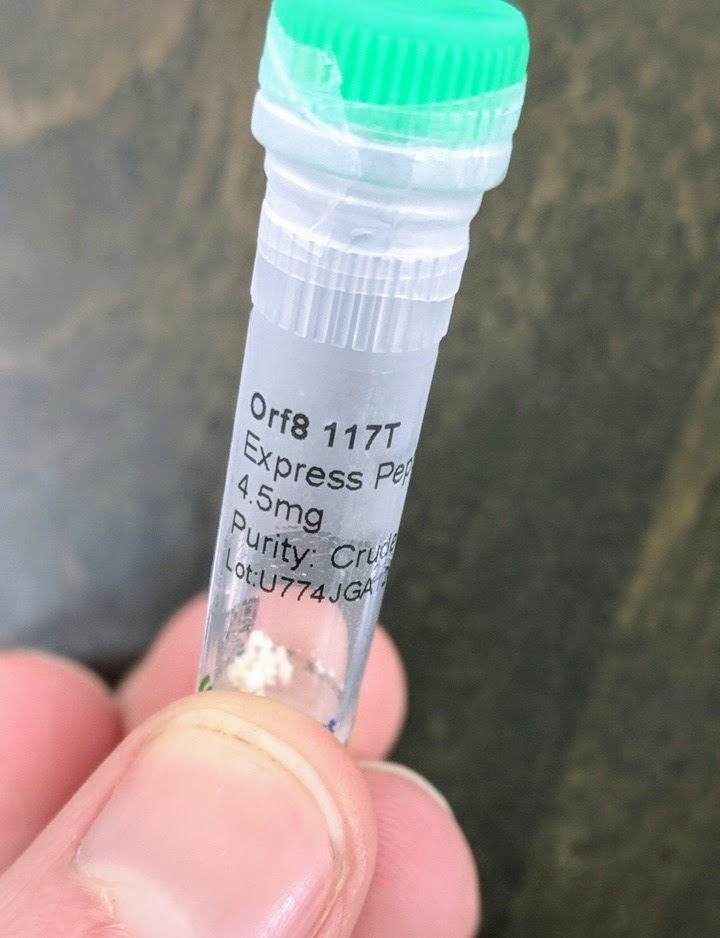
The large majority of the cost (about $850) was the peptides. These are the main active ingredients of the vaccine: short segments of proteins from the COVID virus. They’re all <25 amino acids, so far too small to have any likely function as proteins (for comparison, COVID’s spike protein has 1273 amino acids). They’re just meant to be recognized by the immune system: the immune system learns to recognize these sequences, and that’s what provides immunity.
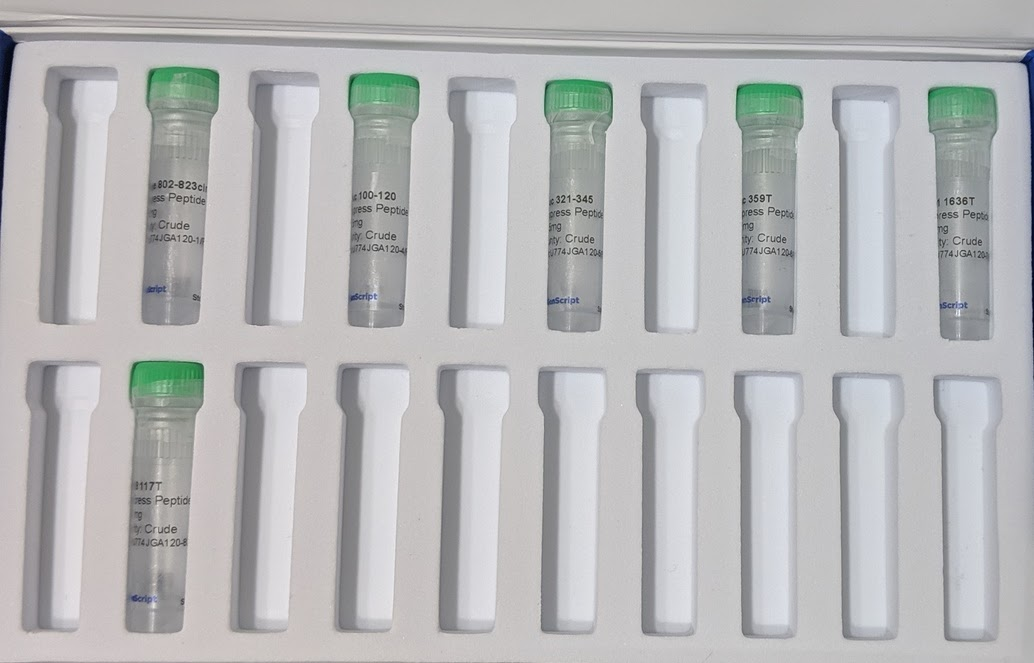
The peptides were custom synthesized. There are companies which synthesize any (short) peptide sequence you want - you can find dozens of them online. The cheapest options suffice for the vaccine - the peptides don’t need to be “purified” (this just means removing partial sequences), they don’t need any special modifications, and very small amounts suffice. The minimum order size from the company we used would have been sufficient for around 250 doses. We bought twice that much (9 mg of each peptide), because it only cost ~$50 extra to get 2x the peptides and extras are nice in case of mistakes.
The only unusual hiccup was an email about customs restrictions on COVID-related peptides. Apparently the company was not allowed to send us 9 mg in one vial, but could send us two vials of 4.5 mg each for each peptide. This didn’t require any effort on my part, other than saying “yes, two vials is fine, thankyou”. Kudos to their customer service for handling it.


Besides the peptides, all the other materials and equipment were on amazon, food grade, in quantities far larger than we are ever likely to use. Peptide synthesis and delivery was the slowest; everything else showed up within ~3 days of ordering (it’s amazon, after all).
The actual preparation process involves three main high-level steps:
- Prepare solutions of each component - basically dissolve everything separately, then stick it in the freezer until it’s needed.
- Circularize two of the peptides. Concretely, this means adding a few grains of activated charcoal to the tube and gently shaking it for three hours. Then, back in the freezer.
- When it’s time for a batch, take everything out of the freezer and mix it together.
Prepping a batch mostly just involves pipetting things into a beaker on a stir plate, sometimes drop-by-drop.

Finally, a dose goes into a microcentrifuge tube. We stick the intake tube of a sprayer into the tube, and inhale.
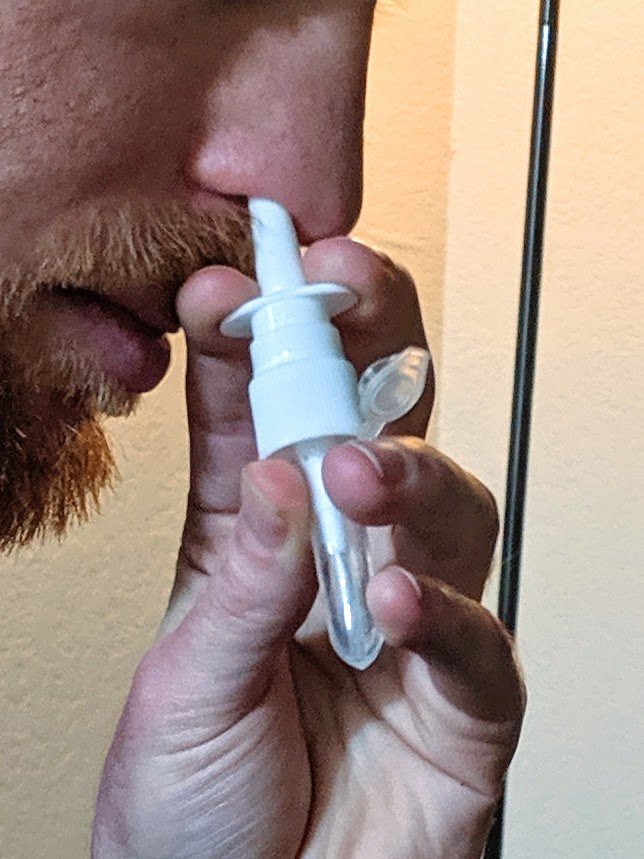
That’s the process, at a high level. Multiple boosters are strongly recommended, so there’s a few iterations of this, though only the “take stuff out of the freezer and mix it together” step needs to be repeated. See the whitepaper for the full protocol details, as well more information about each of the peptides and what the other ingredients do (summary: chitosan nanoparticles).
The Plan
The key problem is how to check that the vaccine worked. If it were injected, that would be easy: just get a standard COVID antibody test. Inhaling makes it a lot harder to hurt yourself, but also complicates testing.
The whitepaper goes into more detail and half-a-dozen different types of immune response, but the basic issue is that immunity response in the mucus lining (i.e. nose, lung, airway surfaces) can occur independently of response in the bloodstream. Commercial COVID antibody tests generally check a blood draw. In principle one can run a similar antibody test on a mucus sample, but <reasons>, so the commercial tests check blood.
(Side note: in many ways immunity in the mucus lining is better than in the blood, since it blocks infection at the point where it’s introduced. This is an advantage of inhaled vaccines over injected. So why do most commercial vaccines inject? Turns out logistics are a major constraint on commercial vaccine design, and injections are surprisingly easier logistically. One of the major relative advantages of radvac is that it’s intended to be prepared on-site shortly before administration, so it can use techniques which work better but don’t scale as well. That largely balances out the constraints of readily-available materials and simple preparation. As usual, the whitepaper goes into much more detail on this, including several other logistics-related relative advantages - multiple boosters, custom peptides, frequent design updates, etc.)
The whitepaper claims that “over a hundred” researchers have self-administered the vaccine so far, but I have not been able to find any data on test results from any of them. The paper says that inhaled vaccine can induce immunity in the blood, but I don’t have a quantitative feel for how likely that is, other than the usual assumption that more dakka [LW · GW] makes it more likely. Meanwhile, I don’t have a convenient way to test for immune response other than the commercial tests.
So, the current plan is to search under the streetlamp [LW · GW]. We’ll just use the commercial tests. Both of us got an antibody test before starting the project, and both came back negative.
My current model is:
- If the vaccine induces an immune response in the blood, then it almost certainly induces one in the mucus lining, but the reverse does not hold. So a positive blood antibody test means it definitely works, a negative antibody test is a weak update against.
- There’s some chance that a few doses are more than enough to induce a blood response.
- There’s some chance that more dakka will induce a blood response, even if the first few doses aren’t enough.
So, we’ll do (up to) two more blood tests. The first will be two weeks after our third (weekly) dose; that one is the “optimistic” test, in case three doses is more-than-enough already. That one is optimistic for another reason as well: synthesis/delivery of three of the nine peptides was delayed, so our first three doses will only use six of them. If the optimistic test comes back positive, great, we’re done.
If that test comes back negative, then the next test will be the “more dakka” test. We’ll add the other three peptides, take another few weeks of boosters, maybe adjust frequency and/or dosage - we’ll consider exactly what changes to make if and when the optimistic test comes back negative. Risks are very minimal (again, see the paper), so throwing more dakka at it makes sense.
Consider this a pre-registration. I intend to share my test results here.
Motivations
Why am I doing this?
I imagine, a year or two from now, looking back and grading my COVID response. When I imagine an A+ response, it’s something like “make my own fast tests, and my own vaccine, test that they actually work, and do all that in spring 2020”. We’ve all been complaining about how “we” (i.e. society) should do these things, yet to a large extent they’re things which we can do for ourselves unilaterally. Doing it for ourselves doesn’t capture all the benefits - lots of fun stuff is still closed/cancelled - but it’s enough to go out, socialize, and generally enjoy life without worrying about COVID.
I’ve written a blog post about Benjamin Jesty [LW · GW], the dairy farmer who successfully immunized his wife and kids against smallpox the same year that King Louis XV of France died of the disease. I explicitly use this as an example of what Rationalism should strive to consistently achieve. Yet when a near-perfect real world equivalent came along, on super-easy mode with most of the work already done by somebody else, it still took me until December to notice. The radvac vaccine showed up in my newsfeed back in July, and I apparently failed to double-click. That level of performance is embarrassing, and I doubt that I will grade my COVID response any higher than a D.
So I’m doing this, in part, to condition the mental motions. To build the habit of Doing This Sort Of Thing, so next time I hopefully do better than a D.
Of course, the concrete benefits are nice too. But at this point it’s only ~4 months until I’d get a vaccine anyway, so the price tag is only arguably worthwhile. It’s still a fun project in its own right, and it gets dramatically cheaper with more people (remember, $1000 bought enough supplies for ~500 doses). Concretely, the largest benefits are in risk reduction. If there’s big hiccups in commercial vaccine deployment, this becomes much more worthwhile. If the South Africa strain turns out to evade commercial vaccines, this becomes much more worthwhile - the radvac design is frequently updated based on the latest COVID research, so we hopefully wouldn’t need to wait around for approval of a new commercial vaccine.
Finally, I'm curious whether it will work - or whether we'll be able to tell that it works. It's a data point as to just how often large bills are left sitting on sidewalks [? · GW] just a little ways off the beaten path.
249 comments
Comments sorted by top scores.
comment by gwillen · 2021-02-03T20:49:53.843Z · LW(p) · GW(p)
Do you have any thoughts on the risks/hazards involved here? To me that's a much more significant consideration than the price. Some thoughts / priors:
- Snorting chemicals I got from the Internet / mixed up myself without really knowing what I was doing: Superficially, seems potentially pretty risky.
- Snorting peptides (assuming that the stuff ordered online was what it claimed to be, was pure and not contaminated with anything hazardous, and that I didn't accidentally create anything hazardous in the process): Definitely not as risky as snorting arbitrary unknown substances. Seems unlikely to be directly poisonous (although that's without reading about the other contents of the vaccine.)
- Snorting COVID-19 peptides, in particular: Should I be worried about things like antibody-dependent enhancement? Are there other possible hazards specific to experimental vaccine administration that I should worry about? I'm sure the paper talks about this stuff, but I'm not a biologist so I can't promise I'd understand it if I read it.
- Is there a possibility that this vaccine is both ineffective, and interferes in some way with the effectiveness of subsequent administration of a different vaccine?
From a risk perspective, the fact that this is intranasal rather than injected makes it feel safer to self-administer, I expect, but is that feeling really justified? Obviously for this vaccine to work, it has to be creating substantial immune effects, at which point I have to ask: what are the risks involved in creating substantial immune effects in my body using a thing I found on the Internet, which has received comparatively very little testing, and without enough knowledge to really verify any of the claims myself?
Replies from: johnswentworth, lincolnquirk↑ comment by johnswentworth · 2021-02-03T21:32:00.207Z · LW(p) · GW(p)
The main answer here is "see the paper"; there's a lot of discussion about this stuff. I'll summarize a few points, as I understand them, relevant to your particular thoughts.
- The "snorting chemicals" aspect is generally not much of an issue, since every ingredient other than the peptides is food-grade, and the quantity in a dose is tiny (one dose is <1 mL, and most of that volume is vinegar and water). If you were eating this stuff in your food and coughed on it, you'd probably get a higher dose than what's in the vaccine.
- Peptide synthesis services generally provide various quality control checks on the product (some free, some upcharge). So at least you'll know what you're getting.
- Antibody-dependent enhancement is one of the main things the paper discusses. It's pretty rare to begin with, and the cases where it's happened have some patterns to them which can be avoided; the peptides are chosen to avoid those pitfalls. Reading between the lines, it sounds to me like historical cases were largely a by-product of historical vaccine techniques (e.g. attaching pieces of one virus to the backbone of another in the case of Dengvaxia) which aren't used here.
- As I understand it, interfering with administration of a later vaccine while also being ineffective would involve basically the same pieces as antibody-dependent enhancement. The paper did not specifically discuss this, though.
From a risk perspective, the fact that this is intranasal rather than injected makes it feel safer to self-administer, I expect, but is that feeling really justified?
Tongue-in-cheek answer: I'm generally pretty comfortable snorting small amounts of things which go in food; this sometimes happens by accident when eating anyway. Injecting small amounts of things which go in food, not so much.
More seriously, all sorts of shit goes into our noses all the time. The blood depends more on being kept separate from the outside world.
[W]hat are the risks involved in creating substantial immune effects in my body using a thing I found on the Internet, which has received comparatively very little testing, and without enough knowledge to really verify any of the claims myself?
This is one of the great challenges of Rationality: you need some level of expertise yourself before you can distinguish real experts from fake [LW · GW]. There is no substitute for learning at least some amount oneself, and thinking through the gears oneself.
Replies from: Dentin, gwillen, cistran↑ comment by Dentin · 2021-02-04T12:24:16.767Z · LW(p) · GW(p)
Regarding the final paragraph, "you need some level of expertise yourself before you can distinguish real experts from fake": that has been the number one reason I didn't beat johnswentworth to the punch and post first with my experience.
I have learned more about biochemistry in the last three months than in my entire prior life combined. It has taken me three months of research, asking questions, and conferring with experts to get sufficient confidence in my understanding to commit to the project.
I'm incredibly thankful to you (johnswentworth) for posting this article; it tracks almost perfectly with my understanding, and I have no significant model conflicts with any of your observations. It raises my confidence in both my understanding, and the project, substantially.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-04T17:35:58.068Z · LW(p) · GW(p)
I'd be very interested in a post on what you learned! I relied mostly on general bio background from undergrad, it sounds like you probably went into more depth in areas specifically relevant to this.
↑ comment by gwillen · 2021-02-03T21:48:09.774Z · LW(p) · GW(p)
Thanks, I really appreciate you taking the time to respond.
I should probably have clarified my current views / epistemic status in my comment, since I think it sounded more skeptical than I actually am. I would say it's something like: "I expect this is quite possibly a good idea, and most probably at worst a neutral idea. I am interested in trying to elicit anything in the long tail of risks that could change that."
(I guess I did also want to encourage other people to at least briefly consider risks before trying this themselves -- although given the complexity and expense, perhaps I shouldn't worry that anybody might rush to try it.)
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-03T21:51:13.849Z · LW(p) · GW(p)
No worries, these are the right questions to ask and I'm glad someone brought them up.
↑ comment by cistran · 2021-02-10T18:23:51.818Z · LW(p) · GW(p)
How many times do you snort adjuvants with your food? I bet the concentration of adjuvants in your homebrew is much higher than what a person could accidentally inhale. This is on purpose of course, so that you are much more likely to get an immune response. But your comparison with things that are not designed to elicit an immune response seems somewhat optimistic.
↑ comment by lincolnquirk · 2021-02-03T21:19:29.286Z · LW(p) · GW(p)
I think a lot of these questions are answered in the radvac paper. I sent a copy of it to a biologist I know, and asked if he thought it was crazy to do this, and he read it and said “geez this looks safer than doing drugs”. I don’t have enough expertise to add anything beyond that.
Replies from: gwillen↑ comment by gwillen · 2021-02-03T21:23:03.244Z · LW(p) · GW(p)
Thanks, that's helpful (and hilarious.) I am looking through the paper now, and it definitely at least purports to answer some of my questions/concerns. (I haven't had a chance to follow the references to see the details.) I would love to hear more takes from people expert enough to weigh in.
comment by Bird Concept (jacobjacob) · 2021-02-08T02:25:23.457Z · LW(p) · GW(p)
Well, this post was just crying out for some embedded predictions! So here we go:
Thanks johnswentworth for help with some of the operationalisations!
I included many different ones, as I think it is often good try to triangulate high stakes questions via different operationalisations. This reduces some some "edge-case noise" stemming from answering vague questions in overly specific ways.
Replies from: jacobjacob, Benito, eillasti, ChristianKl, Pongo↑ comment by Bird Concept (jacobjacob) · 2021-02-09T03:19:19.277Z · LW(p) · GW(p)
@Davidmanheim you're a pretty big outlier here, and this is also the kind of question where I'd trust your judgement a fair bit:

So curious if you wanted to elaborate a bit on your model?
Replies from: Davidmanheim↑ comment by Davidmanheim · 2021-02-09T06:12:43.093Z · LW(p) · GW(p)
First, base rates are critical. Looking at potential drugs overall, the rate of approvals due to safety alone - i.e. "Investigational New Drugs" to phase-II efficacy trials, is very low. Phase 1 trials are typically 80-100 people, and most don't manage to make it past that stage. It would take much stronger evidence than I have seen to think that this vaccine is going to be outside of the norm.
Second, even if the process as done was safe, I can't imagine that greater than 99% of people manage to do this without screwing up in some serious way. That's less true of the LW crowd, but I don't think people are aware of how dumb the mistakes that get made are, or how much quality control matters, and how difficult it is with trying to enforce it for DIY projects.
Lastly, I'm well within the consensus for almost all the rest of the questions - I think it probably works in most cases, and I think it will have side effects in far fewer than 50% of cases.
(But another place I'm a bit outside the consensus is that I think it's unlikely to trigger standard antibody tests, since standard antibody tests are looking for antibodies against a specific part of the virus, and I'm unsure, reading the "Antibodies and B-cell immune response" section of the white paper, that standard tests would detect the elicited types of NABs.)
↑ comment by Raemon · 2021-02-26T01:39:55.901Z · LW(p) · GW(p)
As someone considering taking it, I'd be interested in whether you have models of particular side effects or severities that might happen and why? Do you just have vague "accidents and harmful unknown unknowns aren't that unlikely here"?
I have not currently read the paper or looked into anything very hard. But one question I'd have is "given the ingredients you're working with, is there are particularly obvious way to mix these on purpose that'd result in something harmful happening?"
Replies from: Davidmanheim↑ comment by Davidmanheim · 2021-02-28T09:49:01.126Z · LW(p) · GW(p)
Mostly vague "accidents and harmful unknown unknowns aren't that unlikely here" - because we have data on baseline success at "not have harmful side effects," and it is low. We also know that lots of important side effects are unusual, so the expected loss can be high even after a number of "successes," and this is doubly true because no-one is actually tracking side effects. We don't know much about efficacy either, but again, on base rates it is somewhat low. (Base rates for mRNA are less clear, and may be far higher - but these sequences are unfiltered, so I'm not sure even those bse rates would apply.)
Finally, getting the adjuvants to work is typically tricky for vaccines, and I'd be very concerned about making them useless, or inducing reactions to something other than the virus. But if you want to know about intentional misuse, it's relatively low. I would wonder about peanut protein to induce you to develop a new allergy because you primed your immune system to react to a new substance, but you'd need someone more expert than I.
Overall, I'd be really happy taking bets that in 20 years, looking back with (hopefully) much greater understanding of mRNA vaccines, a majority of immunologists would respond to hearing details about this idea with a solid "that's idiotic, what the hell were those idiots thinking?" (If anyone wants to arrange details of this bet, let me know - it sounds like a great way to diversify and boost my expected retirement returns.)
↑ comment by Ben Pace (Benito) · 2021-02-08T20:36:19.514Z · LW(p) · GW(p)
This was great.
↑ comment by eillasti · 2021-03-02T23:06:39.296Z · LW(p) · GW(p)
> The radvac vaccine will have serious side effects (i.e. besides stuffy nose for a day) for >50% of people who try it
It should be well below 1%. Firstly, if it were that bad as to cause serious side effects for >50% of people who try it, would the RaDVaC team risk promoting it? Secondly, if it were that bad, wouldn’t we hear bad stories about side effects? Thirdly, getting serious side effects accidentally in >50% cases sounds pretty hard on its own.
> The radvac vaccine induces antibodies detectable in a standard commercial blood test in most people, using the dosage in the paper with 2 booster shots
<1%, because RaDVaC team has tried it and didn’t manage to get any positive result.
> The radvac vaccine induces antibodies detectable in a standard commercial blood test in most people, using "more dakka", for some reasonable version of "more dakka"
This greatly depends on what “more dakka” and “reasonable version” means. I assume that “reasonable version” implies "doesn't cause too much harm due to immune system overstimulation”. If “more dakka” means simply a higher dosage, then I think, that this is unlikely (5%), because 1) RaDVaC team experimented on themselves quite a bit, they received a lot of dakka, but no commercial blood test detection, 2) RaDVaC team seems reasonable enough to try this approach if it looked promising. If “more dakka“ includes stronger adjuvants (chitosan is considered a weak, but safe one), then it is much more likely (20%?), because RaDVaC team didn’t investigate those (for a reason) and it sounds plausible that you can get an immune response by irritating the immune system really, really strongly.
Replies from: jacobjacob↑ comment by Bird Concept (jacobjacob) · 2021-03-03T03:28:14.520Z · LW(p) · GW(p)
<1%, because RaDVaC team has tried it and didn’t manage to get any positive result.
That's false, they got several positive anitbody results in ~June or so last year. See a comment elsewhere on this post.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-03-03T05:13:40.802Z · LW(p) · GW(p)
I think he might mean commercial blood antibody tests specifically?
Replies from: eillasti↑ comment by ChristianKl · 2021-02-09T08:57:06.375Z · LW(p) · GW(p)
I would prefer "immunizes against COVID-19" to be better defined when asking the question. Immunizing against becoming systomatic with COVID-19 is a different value then immunizing against not being infectious with COVID-19 and both of those matter. Preventing rate of hospitalization and rate of death would also be important.
Replies from: Davidmanheim↑ comment by Davidmanheim · 2021-02-11T13:41:56.224Z · LW(p) · GW(p)
In epidemiology / medicine, etc. "Immunizes" has a technical meaning - it means you cannot contract or carry the disease. (i.e. not that you don't get symptoms.)
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-11T14:59:06.186Z · LW(p) · GW(p)
By that definition Moderna and BionTech don't have vaccines that are proven to immunize people against COVID-19. That technical meaning might be used in some communities but I think if you would ask the median rationalist whether or not there was a clinical trial whether the Moderna or Pfizer vaccine immunizes people against COVID-19 they would say "yes, there was such a trial".
If a journalist would ask someone at the FDA or CDC the question whether such a trial I doubt they would get the answer "technically there wasn't a trial that showed the those vaccines immunize anyone".
The idea that the technical meaning of a scientific term is something that isn't operationalized seems to me also problematic. There might be people in epidemiology who believe that but it's ontologically problematic and causes a lot of harm.
Replies from: Davidmanheim↑ comment by Davidmanheim · 2021-02-12T14:02:52.785Z · LW(p) · GW(p)
"Immunity" and "efficacy" seem like they should refer to the same thing, but they really don't. And if you talk to people at the FDA, or CDC, they should, and probably would, talk about efficacy, not immunity, when talking about these vaccines.
And I understand that the technical terms and usage aren't the same as what people understand, and I was trying to point out that for technical usage, the terms don't quite mean the things you were assuming.
And yes, the vaccines have not been proven to provide immunizing protection - which again, is different than efficacy. (But the vaccines do almost certainly provide immunizing protection for some people, just based on the obvious prior information and the current data - though it's unclear how well they do so, at how long after the vaccine.)
And, to make things worse, even efficacy is unclearly defined. It gets defined in each clinical trial - differently for each drug/vaccine/etc. and I don't think it actually mean the same thing for the currently approved COVID-19 vaccines. It's pretty similar, stopping symptomatic cases, but even given the same endpoint, it's not necessarily identical, since the studies picked how to measure the endpoints independently, and differently.
comment by JenniferRM · 2021-02-04T02:46:13.353Z · LW(p) · GW(p)
You have my admiration, and my hope that you are calculating the risks accurately!
I have not read the RaDVaC paper so I don't have a good object level model of safety and risks. From a distance it looks like heroism, because from a distance it looks like taking a risk in a way that could provide a role model for many if it works safely! It reminds me a bit of Seth Roberts who was a part of the extended tribe who did awesome stuff over and over again (seemingly safely) but who also may have eventually guessed wrong about safety.
I guess I just want to say: "This is so freaking awesome, and PLEASE be very careful, and also please keep going if the risks seem worth the benefits."
If you get a positive antibody result, have you thought about a personal challenge trial?
The big benefits to be gained from vaccination seem to be to be behavioral: going out, doing life similarly to the Before Times... which is similar to a partial/random/natural sort of "challenge trial".
I wonder if 1daysooner can or would be interested in keeping track of people who have tried the RaDVaC option, to build up knowledge (based on accidental exposures or intentional challenges) of some sort.
comment by DirectedEvolution (AllAmericanBreakfast) · 2021-02-04T03:18:29.062Z · LW(p) · GW(p)
I double-cruxed this article because my "voice of caution" objected to it.
I eventually realized the issue was that part of my decision-making process when I do something weird, potentially risky, or expensive, is to consult with friends and family. Yet I feel that the feedback I would get from them would be so thoughtless, negative, frustrating, and potentially damaging, that it's not worthwhile. And I don't want to ignore this "consult someone first" rule, because it seems like a generally good rule that loses its force if ignored.
However, I do know some specific people who might be good to talk it over with. They're warm, open-minded, very smart, scientifically literate, unconventional, have my best interests in mind, trustworthy, and willing to discuss this kind of stuff at length. My next move is probably not to read the paper, but rather to discuss it with them.
Replies from: JenniferRM, AllAmericanBreakfast↑ comment by JenniferRM · 2021-02-04T17:01:24.299Z · LW(p) · GW(p)
I think what you have done here is re-invented the actual helpful version of a practice whose authoritarian bureaucratic cargo-culted version is called "anonymous peer review".
It is easy (and maybe dangerously wrong) to come to the straightforward conclusion that peer review in general is simply evil bullshit... until one finds the place from which a benevolent truth-oriented human (like oneself) finds a reason to consult with an actual "epistemic peer" as a prudent and socially-embedded response to one's own uncertainty about things one cares about.
↑ comment by DirectedEvolution (AllAmericanBreakfast) · 2021-02-06T02:43:23.917Z · LW(p) · GW(p)
I talked this out with a consultant friend who got his BS in biology. Here's what we came up with.
A conceptual solution would have the following variables, labeled for clarity.
Cost of vaccine = C
- C = (Cost of manufacturing RacVac) ÷ (Doses you'll administer) + (Dollar value to represent cost of unconventionality of the project)
Probably that vaccine provides value = P
- P = (Chance that RadVac works at all) x (Effectiveness if it does work) x (Chance you'll catch COVID before getting vaccinated) x (Chance you'll fail in your execution)
Value that could be provided per person = V
- V = [ (Dollar value of your life) x (Chance you'll die if you catch COVID) + (Dollar value of avoiding a day on a ventilator) x (Chance of serious case of COVID) x (About 14 days on a ventilator) + (Dollar value of avoiding a day of fatigue/anosia) x (Chance of long-term fatigue/anosia) x (Expected length of long-term fatigue) + (Expected number of days out of work) x (Cost of lost work) + (Expected out-of-pocket cost of medical care if you caught COVID)]
- P' = 1 - (Chance you'll transmit it to a particular other person if you catch it) x (Chance they'd have caught it anyway)
- V' = Calculation of V but for another specific person in your life who'd be at risk of COVID if you caught it
If C < P[V + ΣP'V'], it would be worth taking RadVac.
Potential sources for some of these estimates:
- (Chance that RadVac works at all) = (Number of vaccines major pharma companies send to preclinical trials) / (Number of vaccines they put in clinical trials) x 33.4%
- (Effectiveness if it does work) = (Average effectiveness of mRNA vaccines that have been released so far)
- (Chance you'll catch COVID before getting vaccinated), (Chance you'll transmit it to a particular other person if you catch it), (Chance they'd have caught it anyway) = Calculated by adding up your own and other people's activities using the microCOVID risk calculator.
- (Dollar value of a life) = (Dollar value placed on a citizen's life by their federal government)
- (Chance they'll die if you catch COVID) = Hospitalization and death rates by age
- (Chance of lingering effects of COVID) = 52.3%
However, you'd first want to consider if there are other interventions that are even more cost-effective for the same risk factor. For example, if you're still shopping at the grocery store, consider having your groceries delivered for the next six months.
Replies from: kuudes↑ comment by kuudes · 2021-02-06T12:22:41.779Z · LW(p) · GW(p)
I made a little different, simplified take on the matter:
For Radvac to be net useful, it needs that following is true: p(RV prevents Covid)*p(user gets Covid [is exposed to Covid such that it would lead to infection])*p(Covid causes long term harm) > p(RV causes long term harm)
p(RV harm) is currently from the RV paper likely less than 1/10000, cited example is Pandemrix that caused long term harm of narcolepsy with 1/16000 if you had Swedish or Finnish genome. p(Covid harm) is high in old people, where you can die with up to 25% probability, but for most of young people around here long Covid would seem to dominate and that seems to be maybe 1%. Long Covid probability seems to be not well found, and this seems a likely direction for improving decision with better data.
with these presets we get: p(RV prevents Covid)*p(user gets Covid) > p(RV harm)/p(Covid harm) <=> p(RV prevents Covid)*p(get Covid) > 0,0001/0,01 = 0,01
from this, we get 3 inequalities as boundary conditions: (presume scenario where getting Covid is max, that is 100% => prevention needs to be > 0,01; vice versa)
- p(RV prevents Covid) > 0,01
- p(get Covid) > 0,01
- p(RV prevents Covid)*p(get Covid) > 0,01
so with current boundary conditions the key thing to find out with Radvac is how likely it is to cure Covid. This needs to be shown likely to be over 1% or it should not be used unless other boundary conditions can be shown to differ.
An aside: this same calculation applies to all other vaccines, which is why the effort has been put into making sure p(harm from vaccine) is ascertained to be much less than 1/10000. This making sure the vaccine harms the least is about necessary condition for mass vaccinations to be net useful for the participants themselves. This is why we have used 1 year+ for safety testing, which gives us way better and lower prior for vaccine harm than 1/10000. If you get no long term harm from N trial persons, then per succession rule your naive prior is that p(harm) < 1/(N+2).
Replies from: kuudes↑ comment by kuudes · 2021-02-06T12:28:06.804Z · LW(p) · GW(p)
A friend offered that page 7 of white paper could maybe be used to deduce that Radvac would prevent Covid with ~40%.
This would mean the decision boundaries would get to p(Covid)*40% > 0.01 <=> p(Covid) > 0.01/0.40 <=> p(Covid) > 0.025 so then you would need your chance to get Covid to be over 2.5% for the use to be net beneficial.
If we also presume a 80+ year old person who has 25% probability of death given Covid, then it becomes
p(RV works)*p(get Covid)*p(Covid harm) > p(RV harm) <=> p(Covid)*40%25% > 1/10000 <=> p(Covid) > 0.0001/(0.40.25) = 0.001
so for them the chance to get Covid before official vaccination would need to be over 0.001 for it to be net beneficial with these boundary conditions.
comment by Dentin · 2021-02-03T21:54:05.369Z · LW(p) · GW(p)
Speak of the devil. I literally just placed my peptide order a couple of hours ago. My experience (finding supplies, test runs of mixing the solution, safety profile, analysis, etc.) basically matches up with this post.
Thanks a lot for posting it.
comment by Raemon · 2021-02-04T09:52:37.657Z · LW(p) · GW(p)
Note: this post was frontpaged (despite a general policy of not frontpaging covid content) both because a) it seems pretty important, and b) the rationality life lessons seemed pretty timeless.
Replies from: jsteinhardt↑ comment by jsteinhardt · 2021-02-04T19:38:54.276Z · LW(p) · GW(p)
Hmm, important as in "important to discuss", or "important to hear about"?
My best guess based on talking to a smart open-minded biologist is that this vaccine probably doesn't work, and that the author understates the risks involved. I'm interpreting the decision to frontpage as saying that you think I'm wrong with reasonably high confidence, but I'm not sure if I should interpret it that way.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-04T19:51:14.374Z · LW(p) · GW(p)
You should make a top-level comment about this. Chance that the vaccine works and the associated risks are object-level questions well-worth discussing.
In general, frontpage decisions are not endorsements (though I don't know Raemon's thoughts in this particular case), and this comment section is not the place for a debate about frontpaging norms. This is definitely the place to talk about chance the vaccine works and associated risks, though.
Replies from: jsteinhardt↑ comment by jsteinhardt · 2021-02-05T02:18:13.308Z · LW(p) · GW(p)
I don't think I was debating the norms, but clarifying how they apply in this case. Most of my comment was a reaction to the "pretty important" and "timeless life lessons", which would apply to Raemon's comment whether or not he was a moderator.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-05T02:48:57.977Z · LW(p) · GW(p)
Yeah, I don't mean to say your comment was bad as-written, just preemptively heading off a potential thread.
Replies from: jsteinhardt↑ comment by jsteinhardt · 2021-02-05T02:52:45.316Z · LW(p) · GW(p)
Ah got it, thanks!
comment by jsteinhardt · 2021-02-05T02:24:30.096Z · LW(p) · GW(p)
Have you run this by a trusted bio expert? When I did this test (picking a bio person who I know personally, who I think of as open-minded and fairly smart), they thought that this vaccine is pretty unlikely to be effective and that the risks in this article may be understated (e.g. food grade is lower-quality than lab grade, and it's not obvious that inhaling food is completely safe). I don't know enough biology to evaluate their argument, beyond my respect for them.
I'd be curious if the author, or others who are considering trying this, have applied this test.
My (fairly uninformed) estimates would be:
- 10% chance that the vaccine works in the abstract
- 4% chance that it works for a given LW user
- 3% chance that a given LW user has an adverse reaction
-12% chance at least 1 LW user has an adverse reaction
Of course, from a selfish perspective, I am happy for others to try this. In the 10% of cases where it works I will be glad to have that information. I'm more worried that some might substantially overestimate the benefit and underestimate the risks, however.
Replies from: Dentin, johnswentworth↑ comment by Dentin · 2021-02-05T12:28:27.107Z · LW(p) · GW(p)
In my case, yes. My bio expert indicated that it was likely to be effective (more than 50%, but less than 90%) and that the risks were effectively zero in terms of serious complications.
Regarding the food grade versus lab grade question, as well as inaccuracies or mistakes in construction of the vaccine, this was a question I spent a reasonable amount of time on. The TL/DR is that the engineering tolerances are incredibly wide; the molecular weight of the chitosan isn't that important, the mixing rate isn't that important other than it be fast enough, the quantities aren't that important, exact peptide quantities aren't that important etc. A lot of these can be off by not just percentage points, but integer factors, and the result will still be acceptable.
It's also worth pointing out that unless you make serious, significant mistakes that dramatically impair effectiveness, you can always just use "more dakka" to overpower the variations. My plan is to mix each batch independently, such that at least some of the construction variations are expected to cancel. (Also, freezing the final vaccine is likely to impair effectiveness, from what little I've found on the topic.)
Replies from: jsteinhardt, Davidmanheim↑ comment by jsteinhardt · 2021-02-06T01:18:27.894Z · LW(p) · GW(p)
I wasn't sure what you meant by more dakka, but do you mean just increasing the dose? I don't see why that would necessarily work--e.g. if the peptide just isn't effective.
I'm confused because we seem to be getting pretty different numbers. I asked another bio friend (who is into DIY stuff) and they also seemed pretty skeptical, and Sarah Constantin seems to be as well: https://twitter.com/s_r_constantin/status/1357652836079837189.
Not disbelieving your account, just noting that we seem to be getting pretty different outputs from the expert-checking process and it seems to be more than just small-sample noise. I'm also confused because I generally trust stuff from George Church's group, although I'm still near the 10% probability I gave above.
I am certainly curious to see whether this does develop measurable antibodies :).
Replies from: John_Maxwell_IV↑ comment by John_Maxwell (John_Maxwell_IV) · 2021-02-13T03:06:48.860Z · LW(p) · GW(p)
↑ comment by Davidmanheim · 2021-02-08T08:59:51.945Z · LW(p) · GW(p)
"the risks were effectively zero in terms of serious complications"
Your expert said that the risk of putting unfiltered peptide strains into your body was negligible? This claim confuses me.
Did you talk to someone who has a background in immunology, or an infectious disease specialist? (The former seems like the more important type of expertise.) And while this isn't my area of expertise, the claim seems wrong. Having your body develop immune reactions to sequences that aren't the full virus seems potentially really bad - because they could look like other things you don't want your body reacting to.
↑ comment by johnswentworth · 2021-02-08T17:25:07.339Z · LW(p) · GW(p)
You actually put unfiltered peptides into your body all the time. Pretty much any biological material you breath in (e.g. ordinary indoor dust or smelling a flower) contains some.
Replies from: Davidmanheim↑ comment by Davidmanheim · 2021-02-08T20:45:45.549Z · LW(p) · GW(p)
Yeah, putting it in your nose definitely has far fewer risks than putting it in your blood, and that greatly reduces my skepticism. But the set of peptide sequences you naturally encounter is a tiny subspace of the total space of possible proteins, and so I'm less convinced that you might not have side effects.
Replies from: adele-lopez-1↑ comment by Adele Lopez (adele-lopez-1) · 2021-02-09T21:03:54.643Z · LW(p) · GW(p)
Also, having random peptides along with an adjuvant (which triggers an immune response) might be risky even in cases where those random peptides are otherwise completely safe.
↑ comment by johnswentworth · 2021-02-08T17:27:23.049Z · LW(p) · GW(p)
I did not try this test. I had enough bio and physiology background to be confident in my own assessment, though I would not advise others to be similarly confident in my assessment - my background is not legible enough for that.
comment by fluffyclouds · 2021-02-07T14:08:19.249Z · LW(p) · GW(p)
There is another Covid-19 peptide vaccine developed by a Dr. Winfried Stöcker. He injected it into ≥64 volunteers, and the results he published look promising. They show both a good level of IgA, IgG and IgM antibodies and ≥ 94% neutralization for the vast majority of the test subjects. According to him (last paragraph of his blog post), none of the test subjects have reported any relevant adverse symptoms.
He describes the manufacturing in his blog (see translation below):
Man nehme dreimal 15 Mikrogramm rekombinante RBD der S1-Untereinheit (Arg319-Phe541) für eine Person. Als Adiuvans habe ich Alhydrogel von InvivoGen verwendet: Ordentlich durchschütteln und davon 200 Mikroliter mit der Tuberkulinspritze aufziehen. In eine größere Spritze 10 Milliliter Kochsalz aufziehen und die 200 Mikroliter dazugeben, mischen. Davon 500 Mikroliter pro Schuss, mit denen man seine Portion Antigen vermischt. Alles hübsch steril.
I've attempted a translation and added some of my own understanding in [square brackets]. Though I'm a German native speaker, I have zero domain knowledge in this field, so please correct me if anything is wrong:
Take three times 15 μg [three doses of 15 μg per person, spaced days apart - see result data linked above for timing] recombinant RBD in the S1 subunit (Arg319-Phe541) for one person. As an adjuvant I used Alhydrogel by InvivoGen: shake thoroughly and then draw 200 μl with a tuberculin syringe. Using a larger syringe, draw 10 ml saline solution and add the 200 μl [of adjuvant], then mix. Of this mix, use 500 μl per shot and mix it with the antigen [the 15 μg recombinant RBD]. Do all this in a sterile way.
If (!) these instructions are exhaustive, it might be easier, though possibly more expensive to produce than RaDVaC. Googling Arg319-Phe541 suggests that 100 μg (6 doses) / 1000 μg (66 doses) of this RBD can be bought for 310 € (52 € per dose) / 1130 € (18 € per dose), though there may be cheaper offers. I have no clue if the Arg319-Phe541 RBD I found here (230-01102-1000-RB listed on biocat.com and raybiotech.com) would be of the right kind to use in a vaccine.
If you have the relevant domain knowledge to evaluate how complete these instructions are or what other risks and benefits compared to RaDVaC this vaccine may have, I'd really appreciate a comment. Likewise (and especially) if you know where to buy the kind of Arg319-Phe541 RBD used for this vaccine.
If you speak German or are comfortable with DeepL: the current edition (06/21) of the German magazine "Der Spiegel" has some more background on its development on pages 44 - 46: PDF (available for free due to misprints).
Edit 1: Detailed serological results (in English) were posted by Dr. Stöcker in May 2020 when he tested the vaccine on himself first.
Replies from: ChristianKl, caffemacchiavelli↑ comment by ChristianKl · 2021-02-07T14:58:22.251Z · LW(p) · GW(p)
If (!) these instructions are exhaustive, it might be easier, though possibly more expensive to produce than RaDVaC.
You need to work a lot more sterile when injecting then when you spray something in your nose. I'd rather have a few additional steps with less problems if I mess up some of the steps.
Replies from: fluffyclouds↑ comment by fluffyclouds · 2021-02-10T15:29:49.828Z · LW(p) · GW(p)
Good point! I've attempted to expand on this a bit, and list the advantages that each vaccine currently seems to have over the other:
For RaDVaC:
- Extensive Documentation, Whitepaper and reasoning about its development available
- Manufacturing does not require a sterile environment
- Simpler administration
- Has a small community, might be easier to exchange questions and results
- Regularly updated (possibly double-edged - seems very useful to keep up with any variant capable of immune escape, but may (?) make it more difficult to estimate efficacy across vaccine generations)
- Designed to prevent immune escape, may still work when commercial vaccines become less effective (uses 9 - 13 peptides instead of just targeting the spike protein like other vaccines)
- Cheaper ingredients, because the peptides required are shorter
For Dr. Stöcker's Vaccine:
- Test results released so far show very good efficacy and safety (for n=64)
- Known-good dosing regimen available
- Efficacy can be verified using commercial blood antibody tests
- Requires only one peptide (which might be orderable as-is, without custom synthesis) and two passive ingredients
One way to achieve sterility might be to use a self-made glovebox (example tutorial). For extra safety, you could also add an intake fan with a HEPA filter to get a positive-pressure sterile environment, which would still be sterile in case of a small enough leak. Or build a positive-pressure hood - more upfront work, but working in it seems easier.
Personally, the good results and self-verification capability (using antibody tests) would make me prefer Dr. Stöcker's vaccine despite the extra hassle, as long as we knew what kind of Arg319-Phe541 peptide we need for it.
Edit 1: Added / edited the last two points in the RaDVaC list, thanks to feedback by ChristianKl [LW(p) · GW(p)].
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-10T16:05:52.574Z · LW(p) · GW(p)
Has a small community, might be easier to exchange questions and results
Given that this community exists it's likely that they somehow privately share results. It would be really interesting to know more about what's going on in that community.
as long as we knew what kind of Arg319-Phe541 peptide we need for it.
I understand Arg319-Phe541 to mean the subsection of the spike protein that begins with arginine at position 319 and ends with phenylalanin at position 541. At the moment I don't immediately find the sequence with googling but it's worth checking whether 319 is indeed arginine and 541 phenylalanin to check whether this interpretation makes sense.
The problem is that this is 222 amino acids longs which is longer then what the peptide sequencing company sell you so you can't get them the same way you get the peptide you need for the RaDVaC vaccine.
Active-Bioscience seems to sell 100 µg of SARS-CoV-2 Spike Glycoprotein-S1 RBD (319-541, biotinylated) recombinant Protein for 1.150 € which gives you enough doses for two people.
I think RaDVaC has another advantage. It's designed to be difficult for the virus to mutate to get immune to it. Having to change 9 different targets is harder for the virus then just having to change the spike protein and given that every approved vaccine targets the spike protein there's a lot of selective pressure on the virus mutating to get immune to it. The targets are also careful chosen to be conserved and thus less likely to mutate.
Replies from: fluffyclouds, johnswentworth↑ comment by fluffyclouds · 2021-02-10T22:12:42.610Z · LW(p) · GW(p)
Thank you, this is helpful - I've edited the parent post to include some of your feedback.
About the community: the only other place I've found so far is /r/radvac - though pretty dead, it may be useful to find people connected to the community. This page also mentions a Boston RaDVaC group.
I understand Arg319-Phe541 to mean the subsection of the spike protein that begins with arginine at position 319 and ends with phenylalanin at position 541.
This seems right - the RaDVaC white paper has a nicely formatted copy of the S protein on page 40, and it begins with R / arginine at 319 and ends with F / phenylalanine at 541.
I found 230-01102-1000-RB by RayBiotech as a cheaper option. If you buy 100 µg of it at 308 € (includes 19 % VAT) you end up at 52 € per dose / 154 € per person. Or you buy 1000 µg at 1129 €, which is enough for 22 people, it's 18 € per dose / 52 € per person. Again, I have no clue about what you have to look out for when buying this peptide (e.g. format, purity, formulation, ...?) for an injected vaccine, any info on this would be highly appreciated!
Apart from that, the data released on the Stöcker vaccine shows that shots 1, 2 and 3 were on average 10 and 32 days apart. Storing the peptides for this amount of time does not seem to be trivial, see e.g. the storage requirements from RayBiotech for their Arg319-Phe541:
Upon arrival, the protein may be stored for 2 weeks at 4 °C. For long term storage, it is recommended to store at -20 °C or -80 °C in appropriate aliquots. Avoid repeated freeze-thaw cycles.
Maintaining 4 °C sounds doable with a good fridge and a data logging thermometer. -20 °C is more tricky - maybe use a home freezer (*** is specced at ≤ -18 °C) and add a data logger. If it then turns out that it can't reach -20 °C, it might be possible to fix that by modding its internal thermostat somehow. Or have access to a lab freezer, or shell out the big bucks (four figures) to buy one.
I am curious about the storage requirements for RaDVaC and couldn't find out anything specific on the webpage or in the whitepaper. Though the latter does mention some steps that were taken to improve peptide stability, it would be nice to have some info on this.
Replies from: kjz↑ comment by kjz · 2021-02-10T22:48:52.277Z · LW(p) · GW(p)
Maintaining 4 °C sounds doable with a good fridge and a data logging thermometer. -20 °C is more tricky - maybe use a home freezer (*** is specced at ≤ -18 °C) and add a data logger. If it then turns out that it can't reach -20 °C, it might be possible to fix that by modding its internal thermostat somehow. Or have access to a lab freezer, or shell out the big bucks (four figures) to buy one.
As someone who has worked in the labs a long time, I wouldn't worry about having to hit exactly -20 °C; that basically just means "freezer temperature". Lab freezers don't work any differently than home freezers as far as I can tell, although they do have certain safety features that a home freezer wouldn't. But the temperature can still vary a few degrees up or down, and it shouldn't affect your storage much. The (very) general rule of thumb is a difference of +/- 10 °C makes chemical reactions (such as peptide degradation) go 2x faster/slower. So even having to store in a fridge temporarily would only be ~4x faster than a freezer, still maybe good enough for one's purposes.
The big difference comes for -20 °C vs -80 °C, since there you have a 2^6 or 64-fold rate difference. So something that can last for a month at -80 °C might degrade in half a day in a freezer. Hence the complex supply chains needed for such vaccines.
Replies from: translunar↑ comment by Juno (translunar) · 2021-02-17T16:24:17.044Z · LW(p) · GW(p)
Lab freezers have no defrost cycle. Home freezers often do, which prevents ice buildup but also means they don’t maintain temperature.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-17T17:14:27.940Z · LW(p) · GW(p)
So that's why those damn things were always so full of ice! Thankyou, I did not know this before.
↑ comment by johnswentworth · 2021-02-10T16:56:39.681Z · LW(p) · GW(p)
This thread was excellent, strong upvotes for both of you.
↑ comment by caffemacchiavelli · 2021-02-10T20:26:33.729Z · LW(p) · GW(p)
Thanks for posting this, this looks excellent.
It's my impression that you can indeed just buy the antigen needed - the lowest price for 1mg I found was around 900€. Allowing for 10% waste, this would cover 20 individuals at 45µg each. I looked at the antigen test results first and was worried that the vaccine wouldn't perform as well in live tests, but the neutralization results Stöcker posted are quite promising and a Nature study suggests that using 319–545 of the RBD is effective in providing immunity to live virus in primates. I don't expect the remaining amino acids will affect the results in a major way, so this looks quite promising indeed.
I've asked a few people with more domain knowledge to comment on this and depending on their judgment (and access to sterile lab space) I might fund vaccines for myself and friends, given that Germany's vaccine rollout seems to be taking its time.
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-10T20:38:03.150Z · LW(p) · GW(p)
I've asked a few people with more domain knowledge to comment on this and depending on their judgment (and access to sterile lab space) I might fund vaccines for myself and friends, given that Germany's vaccine rollout seems to be taking its time.
Not legal advise: It's my understanding that German law allows you to take things you brew up yourself but only doctors or Heilpraktikers are allowed to give brews like this to other people. If you have a friend who's a doctors or Heilpraktikers who could take the role might be helpful (and that person should have a better idea about the exact conditions under which he can give out brews).
Replies from: caffemacchiavelli↑ comment by caffemacchiavelli · 2021-02-10T20:45:11.390Z · LW(p) · GW(p)
From what preliminary legal advice I've received, I'm allowed to hand it out as a research chemical but anything beyond that might get me into trouble. That sadly limits me to offering it to other nerds who I can reasonably expect to use it for research purposes, but I also highly doubt that anyone outside that cluster would even be interested.
comment by benjaminikuta · 2021-02-04T01:27:38.811Z · LW(p) · GW(p)
I was banned from r/neoliberal for sharing this.
"Spreading dangerous medical information"
Replies from: Yoav Ravid, AI_WAIFU↑ comment by Yoav Ravid · 2021-02-04T05:07:41.612Z · LW(p) · GW(p)
I think deleting it was a fair response (though perhaps banning is a little over the top). assuming the moderator has no way of checking for himself whether this makes sense and he knows he doesn't, he's left with a bet about whether this is the real thing or just bullshit. he expects more bullshit than real things, and he expects the bullshit to be dangerous. so he removes everything that fits this class of things, knowing he might end up also removing something real.
Replies from: Dentin, jimmy, benjaminikuta↑ comment by Dentin · 2021-02-04T12:27:45.849Z · LW(p) · GW(p)
I agree as well. It takes a non-trivial amount of knowledge and research to evaluate the the whitepaper and its claims, and I wouldn't expect the moderator of a "neoliberal" group to have that expertise. We have options with a known risk profile (the commercial vaccines), and there's a spot of fraudulent "cures" out there. The safe thing for a moderator to do is blackhole potentially dangerous claims they don't have the time and/or experience to evaluate.
↑ comment by jimmy · 2021-02-04T19:45:35.463Z · LW(p) · GW(p)
I think this is inaccurately charitable. It's never the case that a moderator has "no way" to know whether it checks out or not. If "Hey, this sounds like it could be dangerous misinfo, how can I know it's not so that I can approve your post?" is too much work and they can't tell the good from bad within the amount of work they're willing to put in, then they are a bad moderator -- at least, with respect to this kind of post. Even if you can't solve all or even most cases, leaving a "I could be wrong, and I'm open to being surprised" line on all decisions is trivial and can catch the most egregious moderation failures.
Maybe that's acceptable from a neoliberal moderator since it's not the core topic, but the test is "When confronted with evidence that they can correctly evaluate as showing them to have been wrong, do they say 'oops!' and update accordingly, or do they double down and make excuses for doing the wrong thing and not update". I don't know the mod in question, but the former answer is the exception and the latter is the rule. If the rejection note was "Medical stuff isn't allowed because I'm not qualified to sort the good from the bad", then I'd say "fair enough". But actively claiming "Spreading dangerous misinfo!" is rarely done with epistemic humility out of necessity and almost always done out of the kind of epistemic hubris that has gotten us into this mess by denying that there's an upcoming pandemic, denying that masks work and are important, and now denying that we can and should dare to vaccinate in ways that deviate from the phase 3 clinical trials. This kind of behavior is hugely destructive and is largely the result of enabled laziness, so it's really not something we ought to be making excuses for.
Replies from: Yoav Ravid↑ comment by Yoav Ravid · 2021-02-05T05:09:55.565Z · LW(p) · GW(p)
I agree i was being charitable. and yes, i was talking specifically about my expectations from a neoliberal forum moderator (if this was a biology or medicine forum i would have higher standards). my point wasn't necessarily that the mod did that out of good epistemics, just that the decision is justifiable, whatever method he actually used to take it. (i don't know the moderator in question either). actually I'd bet the moderator didn't do it with good epistemics, since he overreacted and outright banned him rather than just deleting it.
Replies from: jimmy↑ comment by jimmy · 2021-02-06T19:10:51.513Z · LW(p) · GW(p)
Being as charitable as the facts allow is great. Starting to shy away from some of the facts so that one can be more charitable than they allow isn't.
The whole point is that this moderators actions aren't justifiable. If they have a "/r/neoliberal isn't the place for medicine, period" stance, that would be justifiable. If the mod deleted the post and said "I don't know how to judge these well so I'm deleting it to be safe, but it's important if true so please let me know why I should approve it", then that would be justifiable as well, even if he ultimately made the wrong call there too.
What that mod actually did, if I'm reading correctly, is to make an active claim that the link is "misinformation" and then ban the person who posted it without giving any avenue to be proven wrong. Playing doctor by asserting truths about medical statements, when one is not competent or qualified to do so, getting it wrong when getting it wrong is harmful, and then shutting down avenues where your mistakes can be shown, is not justifiable behavior. It's shameful behavior, and that mod ought to feel very bad about his or herself until they correct their mistakes and stop harming people out of their own hubris. The charity that there is room for is along the lines of "Maybe the line about misinformation was an uncharitable paraphrase rather than a direct quote" and "Hey, everyone makes mistakes, and even mistakes of hubris can be atoned for" -- not justifying the [if the story is what it seems to be] clearly and very bad behavior itself.
Replies from: benjaminikuta↑ comment by benjaminikuta · 2021-02-06T20:23:07.218Z · LW(p) · GW(p)
>The charity that there is room for is along the lines of "Maybe the line about misinformation was an uncharitable paraphrase rather than a direct quote"
For what it's worth, it was a direct quote, and the entirety of the ban message, other than a link to the comment.
↑ comment by benjaminikuta · 2021-02-04T08:37:01.972Z · LW(p) · GW(p)
Even with a Harvard professor as an author?
Replies from: Yoav Ravid, ChristianKl↑ comment by Yoav Ravid · 2021-02-04T09:23:12.527Z · LW(p) · GW(p)
It depends more on the ignorance of the moderator and on how much time he's willing to spend than on the quality of the evidence. there definitely are cases of of PHDs and maybe even professors advancing pseudoscience. so this doesn't guarantee trustworthiness.
the moderator has to make a decision in a state where he can't trust himself to distinguish real stuff from bullshit. he goes for minimizing harm at the cost of deleting novel good ideas. seems like a sensible decision to me.
Replies from: benjaminikuta↑ comment by benjaminikuta · 2021-02-05T01:59:36.422Z · LW(p) · GW(p)
How often do scholars of such prominence promote dangerous pseudoscience?
Replies from: jsteinhardt, Yoav Ravid↑ comment by jsteinhardt · 2021-02-05T02:16:28.006Z · LW(p) · GW(p)
Often, e.g. Stanford profs claiming that COVID is less deadly than the flu for a recent and related example.
Replies from: waveBidder, cistran↑ comment by waveBidder · 2021-02-06T05:11:33.160Z · LW(p) · GW(p)
John Ioannidis, of all people, who should know better.
↑ comment by Yoav Ravid · 2021-02-05T05:16:42.003Z · LW(p) · GW(p)
Interesting question, i don't know. but it also doesn't matter here. if the moderator is ignorant he's also ignorant of this fact, and thus cannot take it into account or will have to spend effort finding a good answer for it - so we're back at square one.
Replies from: Dentin↑ comment by Dentin · 2021-02-05T12:20:37.533Z · LW(p) · GW(p)
Again, I have to disagree - misinformation is much more likely than information by default, and the moderator need only have a reasonable low-probability prior in order to reject unusual/uncommon claims without evidence.
Replies from: Yoav Ravid↑ comment by Yoav Ravid · 2021-02-05T12:49:14.404Z · LW(p) · GW(p)
I agree with that. not sure what you think i meant that you disagree with it.. (or was it directed at the comment above me?)
Replies from: Dentin↑ comment by Dentin · 2021-02-06T00:15:05.107Z · LW(p) · GW(p)
Sorry about that; I believe I misread your comment as implying that if the moderator is ignorant, he won't have enough information to form a reasonable prior. My disagreement was along that line, as it seems that misinformation, especially about medical things, is so prevalent that everyone's default prior should be 'fraud unless lots of evidence points the other way'.
↑ comment by ChristianKl · 2021-02-04T19:05:11.564Z · LW(p) · GW(p)
I think there's a difference here between sharing a link to RadVac and sharing a link to a LessWrong post by someone without credentials doing something on their own.
↑ comment by AI_WAIFU · 2021-02-05T06:15:55.461Z · LW(p) · GW(p)
I wouldn't look too deeply into that. The selection process for moderators on reddit is essentially first come first serve + how good are you at convincing existing moderators you should join the team. As far as I can tell this process doesn't usually select for "good" moderation, especially once a sub gets big enough that network effects make a subreddit grow despite "bad" moderation. This applies for most values of "good" and "bad".
comment by Czynski (JacobKopczynski) · 2021-02-05T19:45:35.768Z · LW(p) · GW(p)
Via Sarah Constantin's Twitter:
I looked into this, because yay citizen science. I could not find one research study using any of the peptides in the RADVAC white paper that found they inhibited SARS-CoV-2 infection in cells, let alone animals or humans.
and
“Take a random peptide that has never been tested on any living thing” is not at all the same thing as “take a well-known, well-studied recreational drug”, as far as risk goes.
She doesn't explicitly state that this has never been tested on any living thing. Possibly because she wasn't confident enough in her research survey to claim that, possibly because she was drawing a starker contrast than applies to this instance. But all the COVID testing for RADVAC is purely in silico, so while the chemicals involved may be studied for safety in vivo, efficacy is completely untested even at the (much simpler than organism) cell level.
So the EV of the benefits are low, and the risks are unclear.
Replies from: kjz↑ comment by kjz · 2021-02-06T00:06:31.502Z · LW(p) · GW(p)
This article from July 2020 claims that George Church and many of his colleagues had already self administered their vaccine at that point. It's almost certainly true that there hasn't been a clinical trial, because nobody has ever had an incentive to run a clinical trial. I don't think their intent was to publicize this widely or profit commercially from it. Rather, they realized they could just do it, went ahead and did it, and wrote up their findings publicly but under the radar, so other like-minded individuals could duplicate their procedure at their own risk. Remember that they are an academic research group and they face very different incentives than the drug companies trying to vaccinate the general public. In any case, it seems clear that these vaccines have been tested on many living things, just not in an official study.
Replies from: JacobKopczynski↑ comment by Czynski (JacobKopczynski) · 2021-02-06T03:20:56.272Z · LW(p) · GW(p)
No one said anything about a clinical trial. Emphasis added:
I could not find one research study using any of the peptides in the RADVAC white paper that found they inhibited SARS-CoV-2 infection in cells,
Researching the effects in cells requires no IRB approval and publishing the results of that research as a publicly-accessible preprint is not hard. This should be fairly easy to do, for someone with access to a good lab, personal-scale funding, and motivation. I have to assume that Church et. al. have the first two, so either they don't care enough to bother, or they did but the results weren't encouraging (and either kept quiet or just unnoticed). Neither is what I'd call a 'good sign'.
Replies from: kjz↑ comment by kjz · 2021-02-06T17:14:44.409Z · LW(p) · GW(p)
Agree neither Sarah or you had explicitly mentioned a clinical trial. I was pushing back more against Sarah's statement “Take a random peptide that has never been tested on any living thing” and your statement "She doesn't explicitly state that this has never been tested on any living thing", which I interpreted as endorsing the claim that this vaccine has never been tested on any living thing. My point is that there is evidence this vaccine has been tested in living things, namely the humans who claim to have self administered it. I have no strong reason to doubt they have done so, and I haven't seen any reports of harm coming to these individuals as a result (although admittedly I have no idea if such reports would be publicly available). When I mentioned clinical trials, I was trying to think of what evidence might convince Sarah this approach is not as risky as she fears, and a clinical trial was the first thing that came to mind.
This should be fairly easy to do, for someone with access to a good lab, personal-scale funding, and motivation. I have to assume that Church et. al. have the first two, so either they don't care enough to bother, or they did but the results weren't encouraging (and either kept quiet or just unnoticed).
Agree they almost certainly have the first two, but I don't see why they would have had motivation to perform the kind of cell-based studies you are looking for. Here is how I imagine their motivation and incentives throughout the last year, mostly drawn from the article I linked above and info from the radvac website:
- They see Covid is becoming a pandemic, estimate that a commercial vaccine is >1 year away, and wonder if they can develop an open source vaccine that will provide some level of protection more quickly. At this point, their strongest motivation is to develop a vaccine for their own personal use.
- They design the radvac vaccine, and based on their personal and collective understanding of vaccines, biochemistry, immunology, etc., each individual decides it is in their personal best interest to self administer the vaccine.
- They are torn between competing desires to make their protocol and the underlying research public, and to avoid unnecessary attention from regulatory authorities. From the article:
Given the international attention on covid-19 vaccines, and the high political stakes surrounding the crisis, the Radvac group could nevertheless find itself under scrutiny by regulators. “What the FDA really wants to crack down on is anything big, which makes claims, or makes money. And this is none of those,” says Church. “As soon as we do any of those things, they would justifiably crack down. Also, things that get attention. But we haven’t had any so far.”
- Therefore they settle on the strategy of publishing the white paper under the radar, so it is publicly available but attracts as little attention as possible. (With great success I might add, since we are only having this discussion 6 months later!)
- Each individual has already made the decision to self administer based on their personal risk-benefit analysis, without the need for cell-based studies.
- Publishing additional cell-based studies could increase the chance of drawing unwanted regulatory attention to their effort.
- Thus, they don't have strong incentives to carry out any cell-based studies (which would also take time and effort away from higher priority things they might work on instead), and they likely do have incentives to avoid publishing any cell-based studies.
Which leaves us in the current equilibrium where there are no published cell-based studies.
I think your claim that "they don't care enough to bother" is not very accurate, and a consideration of their incentives as I outlined above provides an alternative reason why we might not expect to find any published cell-based studies.
At the end of the day, we all still have to make personal decisions based on the information at our disposal, as incomplete or challenging to interpret as it may be.
Happy to hear any additional thoughts on this topic!
comment by Daniel Kokotajlo (daniel-kokotajlo) · 2021-02-04T17:44:59.236Z · LW(p) · GW(p)
Related: This was discussed on LW in August 2020, someone claims to have done it in December: https://www.lesswrong.com/posts/62WuBbQpSwAbctGDP/what-price-would-you-pay-for-the-radvac-vaccine-and-why [LW · GW]
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-04T20:06:17.298Z · LW(p) · GW(p)
I'm afraid I'm not flexible enough to kick myself that hard.
Replies from: Dentincomment by DirectedEvolution (AllAmericanBreakfast) · 2021-02-04T04:36:10.941Z · LW(p) · GW(p)
Vaccines that are brought to clinical trials have a 33.4% approval rate, which seems like a reasonable estimate of the chances that this vaccine works if executed correctly. Note that this is from trials conducted from 2000-2015.
I probably have a roughly 5% chance of catching COVID before I'm vaccinated. Given my age, COVID would put me at a 0.2% risk of death. Let's double that to account for suffering and the risk of long-term disability.
If I value my life at $10,000,000, then an intervention that gives me a 33.4% chance of avoiding a 5% chance of a 0.4% chance of death is worth $668. So it seems like I'd want to be vaccinating at least one other person in order for this to be worthwhile.
I welcome any further thoughts on this expected value calculation. In particular, I think it's possible that I'm dramatically underestimating the risk and potential severity of long-term symptoms. It doesn't take much additional risk to make this project worthwhile for a single person.
Replies from: Dentin, JacobKopczynski, waveBidder, cursed, None↑ comment by Dentin · 2021-02-04T12:50:41.166Z · LW(p) · GW(p)
Regarding the 33.4% approval rate: based on what I've learned about traditional vaccine development and production in the last few months, I am not at all surprised. Both peptide and RNA vaccines are effectively "state of the art" technologies compared to traditional vaccine techniques. It's like comparing modern non-invasive out-patient surgery to the 1970's equivalent.
You need look no further than the russian and chinese vaccines - those use the rather crude technology of "throw big chunks of inactivated virus particles at the immune system and hope that the immune system guesses the right antibodies to deal with the live version."
Both peptide and RNA vaccines are instead, "we have identified very specific antibodies which we know are effective both from the serum of recovered patients and from computational modeling, then use exactly the minimal protein sequences needed to generate those antibodies."
Both the russian and chinese vaccines use chunks of proteins that are thousands (and likely tens of thousands) of amino acids long, in a mostly inactivated form. The immune system has no idea what to latch onto, what will be effective at stopping replication, but it does generate a bunch of randomish antibodies anyway just in case. In a lot of people, this is enough to take the edge off getting stick.
The peptide and RNA vaccines on the other hand, are extremely narrow. The radvac vaccine for example targets 9 specific virus protein sequences, the shortest of which is 10 amino acids long, and the longest of which is 25. Each of these sequences is from an empirically likely effective antibody, found in real people who have really recovered.
Like I said, very, very different technologies.
Replies from: ChristianKl, Creutzer↑ comment by ChristianKl · 2021-02-04T19:33:23.901Z · LW(p) · GW(p)
Each of these sequences is from an empirically likely effective antibody, found in real people who have really recovered.
That seems to me like a strange statement. In what way are amino acids sequences in the peptides "from antibodies"?
Replies from: TheSimplestExplanation↑ comment by TheSimplestExplanation · 2021-02-04T19:43:46.686Z · LW(p) · GW(p)
It's my impression that the peptides in question are the antigens to those particular antibodies.
Replies from: Dentin↑ comment by Dentin · 2021-02-04T22:37:23.093Z · LW(p) · GW(p)
Yes; sorry I was unclear. Those peptides generate the antibodies we care about, that are known to be effective against the full virus.
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-04T23:51:29.422Z · LW(p) · GW(p)
Those peptides generate the antibodies we care about, that are known to be effective against the full virus.
It's unclear to me to what extend we know this and your description looks to me like it asserts that we know things that are very hard to know.
Replies from: Dentin, TheSimplestExplanation↑ comment by Dentin · 2021-02-05T11:36:12.435Z · LW(p) · GW(p)
A lot of people have been working really hard for the last year to discover, understand, and know these things. It's the foundation for how the mRNA vaccines work.
Perhaps take a look through this:
https://www.sciencedirect.com/science/article/pii/S2319417020301530
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-05T23:33:11.589Z · LW(p) · GW(p)
I seems my intuition is well-founded here. According to Sarah Constantin the peptide here are selected in silico and not based on antibodies developed by infected people.
Replies from: Dentin↑ comment by Dentin · 2021-02-06T00:20:26.456Z · LW(p) · GW(p)
Sarah Constantin is confused, and likely has not spent significant time reviewing the vaccine design. From page 32 of the whitepaper:
"Empirical evidence should dominate selection criteria. Here are some best types of evidence:
- Mapping of epitopes in blood and other samples collected from convalescent patients (ideally stratified by severity of illness). This can be accomplished by a few primary means:
- 3D structural studies and modeling of neutralizing antibody binding to a viral antigen (e.g. Spike protein)
- Mapping of linear B-cell epitopes by binding antibodies in convalescent sera to a library of peptides representing viral antigens. A strong signal in a linear epitope mapping study does not guarantee that the epitope peptide in the context of a vaccine will trigger the production of an antibody that binds to this epitope within the context of the virus. However, it is a good indicator that this is at least possible. Peptides can be constrained to approximate native conformation, making it more likely to bind the native epitope.
- Mapping of T-cell epitopes by stimulating convalescent T-cells with epitope peptides, and measuring their response (e.g. cytokine secretion; ELISpot)
- Epitope peptides from a peptide vaccine that has shown protection against
infection - Successful use of epitope peptides in vaccines that elicit antibodies (or serum)
effective in virus neutralization assays. B-cell epitopes that allow antibody binding
to the virus but don’t block viral function might increase risk of
antibody-dependent enhancement. - Mapped epitopes that are effective in virus neutralization assays (e.g. peptides
compete with viral sequences in cellular infection assays). - Successful use of epitope peptides in vaccines that elicit T-cell responses, or
peptides shown to stimulate T-cells or cytokine production in ELISpot or other
T-cell assay in cells from convalescents."
↑ comment by ChristianKl · 2021-02-06T11:53:42.407Z · LW(p) · GW(p)
Speaking about what are the best types of evidence is different from demostrating that this evidence exists for individual sequences.
If we start with the list the first is Spike 802-823cir. They provide no citations to papers for this and changed the structure in a way the believe to be benefitial (likely based on in silico modelling).
Replies from: jmh↑ comment by jmh · 2021-02-09T00:26:00.537Z · LW(p) · GW(p)
Doesn't this speak to your concern:
Spike 802-823cir: FSQ c LPDPSKPSKRSF c EDLLF ( Cys4, Cys17 disulfide)
IN TESTING, vaccine Generations 5, 6, 7, 8. 9
To preserve the loop structure present in the native conformation, we substituted cysteines for amino acids 4 (Ile>Cys) and 17 (Ile>Cys).
They perform the substitution to keep the shape that our immune system is looking for by recreating a disulfide bond that to form a loop with the same sequence the B-cells are targeting in the virus.
While I agree their expression was "potentially beneficial" (or close) it seems clear to me the point was our B-cells are bonding to that loop and if there are not other aspect in the larger peptide that lead the cell to that site for bonding, construction the loop via the disulfide bond they introduce logically should result in triggering an immune response.
I'm not sure why they would need to provide some type of citation for this, much less that they would even have a source for this specific application.
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-09T00:56:56.677Z · LW(p) · GW(p)
The argument about the substition of the amino acids looks to me like it rests completely on in silicio modeling.
logically should result
That's theory-based reasoning and not empirical evidence based. Sarah Constantin says that everything is theory-based reasoning (supported by computer modeling) and Dentin argues that they not only do theory-based reasoning but also have empiric evidence for individual peptides.
Replies from: FireStormOOO↑ comment by FireStormOOO · 2021-02-18T23:35:37.188Z · LW(p) · GW(p)
While it does seem there was a certain amount of shotgun aproach following a few different lines of reasoning, that critism is difficult to square with actually reading the paper. It looks like the peptide selection was largely empirical and cited. The decisions about how to actually package that info into a vacine is largely educated guesswork (as you say theory, supported by computer modeling).
"Mapping of linear B-cell epitopes by binding antibodies in convalescent
sera to a library of peptides representing viral antigens. A strong signal in a
linear epitope mapping study does not guarantee that the epitope peptide
in the context of a vaccine will trigger the production of an antibody that
binds to this epitope within the context of the virus. However, it is a good
indicator that this is at least possible."
Or as I understood from elsewhere: present antibodies from recovered people to every possible short peptide sequence and see which ones they actually attacked. Make the inference that people with less severe infection had better antibodies than those with more severe symptoms in the event antibodies differed. Package a selection of promising looking pepties into a vacine; choose enough that there's likely multiple effective peptides even if 2/3rds of the choices are duds.
↑ comment by henryaj · 2021-02-06T13:55:15.725Z · LW(p) · GW(p)
I also don't understand her comments about the peptide 'not neutralising COVID in cell [culture]' - why would it? The peptide is just an antigen to get the body to raise an immune response; on its own it doesn't kill COVID.
Replies from: translunar, johnswentworth↑ comment by Juno (translunar) · 2021-02-17T16:28:03.680Z · LW(p) · GW(p)
I interpreted this to mean antibody against the peptide.
↑ comment by johnswentworth · 2021-02-06T16:38:41.127Z · LW(p) · GW(p)
I was also confused about that. I'm sure some kind of cell-culture method is useful for testing vaccines, but I don't know exactly what's involved. Just culturing immune cells, maybe?
↑ comment by TheSimplestExplanation · 2021-02-05T10:39:04.962Z · LW(p) · GW(p)
Do you have doubts? It seems plausible to me?
↑ comment by Creutzer · 2021-02-06T19:53:43.864Z · LW(p) · GW(p)
Both the russian and chinese vaccines use chunks of proteins that are thousands (and likely tens of thousands) of amino acids long, in a mostly inactivated form.
The Russian vaccine, unlike the Chinese one, is not an inactivated virus. It uses an adenovirus vector for delivery of genetic material that makes the body's cells synthesise antigen material, much like the AstraZeneca/Oxford vaccine.
↑ comment by Czynski (JacobKopczynski) · 2021-02-05T20:03:00.905Z · LW(p) · GW(p)
Vaccines that are brought to clinical trials
This is an inappropriate reference class. This has no in vitro testing conducted; it's entirely a computational model. "“Peptide” just means “sequence of amino acids.” Would you conclude that, because some lines of code can navigate a rocket to the moon, that your code is pretty likely to navigate a rocket to Mars?". A vaccine brought to clinical trials has already overcome many more hurdles than this has. Generally in vitro testing (I think both for safety and efficacy), in vivo safety testing (in rats!), and some scaled-up testing in other animal models.
This isn't a vaccine candidate. This is a promising research lead for a vaccine candidate.
Replies from: johnswentworth, AllAmericanBreakfast↑ comment by johnswentworth · 2021-02-05T20:20:17.706Z · LW(p) · GW(p)
A vaccine brought to clinical trials has already overcome many more hurdles than this has.
I'm not sure what your level of background knowledge is, but I heard that the Moderna vaccine was designed in two days. Clearly they did not do any significant in vitro or in vivo testing in that timespan. Maybe they did some in-vitro before human trials, I don't know; that would support an argument against using "vaccine brought to clinical trials" as a reference class.
But the deeper point which this is trying to operationalize is "vaccine design just isn't that hard", in the sense that we don't need to test many designs to find one which works. People basically-know-how-to-design-vaccines, maybe not to quite the same extent as people basically-know-how-to-design-bridges, but to enough of an extent that experimental verification just isn't necessary in order to get a >50% chance that the design works, especially for relatively-mechanical designs like mRNA or peptides.
Under this view, the reasons we don't see nearly-every vaccine trial succeed are (1) commercial vaccines are harder than lab (especially if you want no boosters, easy logistics, etc), and (2) diseases which are harder-than-average will naturally end up with disproportionately many trials, and (3) out-of-date companies take time to die off.
Replies from: JacobKopczynski, CharlieMarr@protonmail.com↑ comment by Czynski (JacobKopczynski) · 2021-02-06T03:10:34.328Z · LW(p) · GW(p)
The in vitro testing had already been done before those two days; they had the basic structure of the vaccine known, so once they had a virus sample they could fill in the blank (the spike protein of this particular virus rather than another in its 'family') with high confidence that it would work. One of the two days, IIUC, was spent synthesizing a sample vaccine and running some very-short-term tests.
This, again, has only simulation to support that it's hitting the correct target at all. There is no indication that any of that has been done, by the authors or anyone else; IIUC there is not a clear path for doing short-term tests for this type of vaccine.
Also, it's not my background knowledge that you should be comparing to, it's Sarah's. And I literally believe there is no one in the world who can be more trusted to reason clearly, well-informedly, correctly, and with humility and arrogance in their respective correct places than Sarah Constantin. Evaluating biomedical research has been her job for many years, with some gaps, and she's really good at rationality, Aumann-level good. The bare fact that Sarah C thinks this is very unlikely to work is conclusive on its own.
↑ comment by CharlieMarr@protonmail.com · 2021-02-26T14:50:55.617Z · LW(p) · GW(p)
How does the "vaccine design just isn't that hard" align with these points?
a) Average time to develop a vaccine for a new virus is many years
b) There is still no HIV vaccine after 35 years of well-funded research
c) Until a few months ago, there were no approved coronavirus vaccines for humans
I'm prepared to accept that "bureaucracy" is the main cause for the delays in standard big company vaccine development and approval.
But if it's easy to develop vaccines, why has there been no coronavirus vaccine previously? Why is there still no vaccine for SARS 1 or MERS or the common cold? Why was this Radvac idea or something similar not rolled out pre-Covid? (or was it? maybe nasal vaccines are easier?)
Anyway, I'm just stuck on the logical conflict between "it's easy to develop a coronavirus vaccine" and "we've never had one (approved) before." Any thoughts?
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-26T18:53:00.316Z · LW(p) · GW(p)
Good questions.
First, I expect a disproportionate number of vaccine trials are for "unusually difficult" viruses, like HIV. After all, if it's an "easy" virus to make a vaccine for, then the first or second trial should work. It's only the "hard" viruses which require a large number of trials.
But if it's easy to develop vaccines, why has there been no coronavirus vaccine previously? Why is there still no vaccine for SARS 1 or MERS or the common cold? Why was this Radvac idea or something similar not rolled out pre-Covid?
I expect this is still mainly a result of regulatory hurdles. Clinical trials are slow and expensive, so there has to be a pretty big pot of gold at the end of the rainbow to make it happen. Also, companies tend to do what they already know how to do, so newer methods like mRNA or peptide vaccines usually require a big shock (like COVID) in order to see rapid adoption.
↑ comment by DirectedEvolution (AllAmericanBreakfast) · 2021-02-05T20:42:02.026Z · LW(p) · GW(p)
I agree with the point of your comment, that vaccines brought to clinical trials is a suboptimal reference class. However, I think that this is a locally invalid argument:
Would you conclude that, because some lines of code can navigate a rocket to the moon, that your code is pretty likely to navigate a rocket to Mars?
A computational model plus grounding in theory, if done right, should increase our confidence in the the efficacy of a sequence of peptides taken from the virus above the efficacy we'd assume for a random sequence of peptides.
How much? Can't say.
As others have pointed out here, we on the other hand are comparing a new and perhaps much more effective means of designing a vaccine to the methods that were used from 2000-2015, which may be less effective. Hence, perhaps the reference class is suboptimal in the opposite direction as well.
I have no way to know how to weigh these competing factors. So I think the best thing to do is to start with the basic formula I concocted above, then modify it based on our intuitions about these other factors.
Alternatively, you could very justifiably stick with the rule "I don't take untested medications." Although as someone else pointed out, if you have that rule then perhaps you should also make sure to not use any drugs? I don't have the answer, but wanted to try and provide some clarity for people who are considering breaking the "take no untested medications" rule.
↑ comment by waveBidder · 2021-02-04T07:21:51.175Z · LW(p) · GW(p)
You're missing the very real possibility of long-term negative side-effects from the vaccine, such as triggering an auto-immune disease or actually increasing your susceptibility, both mentioned in the whitepaper (whose risk-assessment I would be pretty sceptical of). I would think of this as more a trade-off between risks of side effects and COVID risks, rather than whether or not you can afford it.
Replies from: Dentin↑ comment by Dentin · 2021-02-05T12:31:50.216Z · LW(p) · GW(p)
Yes. The differential tradeoff is how one should evaluate this. The only reason my evaluation came out in favor of trying the radvac vaccine is because I have a high-risk event coming up in the next few months, and I am extremely unlikely to be able to acquire a commercial vaccine before then.
↑ comment by cursed · 2021-02-04T07:18:37.859Z · LW(p) · GW(p)
Vaccines that are brought to clinical trials have a 33.4% approval rate, which seems like a reasonable estimate of the chances that this vaccine works if executed correctly.
I don't follow. Don't vaccines have trials on cells, mice, primates, before clinical? So unless radvac has also done similar testing, this 33.4% isn't comparable.
↑ comment by [deleted] · 2021-02-04T06:41:24.542Z · LW(p) · GW(p)
Do you value your life at ten million? As in, would you take a 50% chance of death for five million? If so, why are you not smuggling drugs or whatever?
Replies from: AllAmericanBreakfast, Bucky, cistran↑ comment by DirectedEvolution (AllAmericanBreakfast) · 2021-02-04T16:46:13.943Z · LW(p) · GW(p)
Well, a couple of researchers estimated that drug mules made a median of $1313 back in 2014, so I'd need to smuggle a lot of cocaine to earn that much. Seems like it would take a while...
↑ comment by Bucky · 2021-02-04T13:44:24.062Z · LW(p) · GW(p)
Say my life expectancy from now is 50 years and I work at an hourly salary of $30 (~$60k yearly salary) then I implicitly value the remaining 310,250 hours of my waking life at something like $9.3m total. This breaks down if offered larger probabilities of death and larger amounts of money (e.g. opportunity cost) but $10m seems like a sensible place to start for a Fermi calculation.
In this case we don't even have to worry about larger probabilities of death - the calculation here is essentially an expected gain of 1.2 days of life for $1000 which comes to about $50 per hour of waking life. Instead of making a vaccine only for myself I would be better just to take half a week unpaid leave and gain the same amount of time for a cost of only $600.
Obviously this has some assumptions baked in (like that I don't value my job) but If It’s Worth Doing, It’s Worth Doing With Made-Up Statistics.
comment by Sniffnoy · 2021-02-04T06:12:25.675Z · LW(p) · GW(p)
Wow!
I guess a thing that still bugs me after reading the rest of the comments is, if it turns out that this vaccine only offers protection against inhaling the virus though the nose, how much does that help when one considers that one could also inhale it through the mouth? Like, I worry that after taking this I'd still need to avoiding indoor spaces with other people, etc, which would defeat a lot of the benefit of it.
But, if it turns out that it does yield antibodies in the blood, then... this sounds very much worth trying!
Replies from: Dentin↑ comment by Dentin · 2021-02-04T12:34:38.182Z · LW(p) · GW(p)
My understanding is that it helps a lot.
The biggest benefit seems to be that the immune system is primed in at least some fashion; it knows what to look for, and it has readily available tools that should be effective. It doesn't have to take a day or a week to try random things before it finally discovers a particularly effective antibody and gets the production chain ramped up to start a proper immune response.
Instead, your immune system will very quickly get a signal it understands as bad and can immediately start ramping up when it does detect the virus.
Keep in mind that the commercial vaccines don't have 100% success rate in that some people still get sick, but the 'priming' of the immune response is still there. I believe this is why the death rate / severe complications rate is effectively zero for immunized patients, even though it's possible to get sick.
(Again, my understanding. I would very much appreciate correction/clarifications here.)
comment by AnnaSalamon · 2021-02-04T01:52:48.698Z · LW(p) · GW(p)
Neat! Will you also use try commercial antibody tests on your mucus, or is that known to not-work?
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-04T02:21:16.453Z · LW(p) · GW(p)
The way the tests we've used work is a nurse takes a blood draw, then ships it off somewhere for the antibody test, so there's not really an opportunity to use mucus in the test. If the next test comes back negative, then I might search harder for an at-home antibody test and a protocol for mucus.
Replies from: jmh↑ comment by jmh · 2021-02-04T20:17:17.691Z · LW(p) · GW(p)
Would an alternative (and possibly easier) approach be to simply take some additional doses -- it's my understanding that you really cannot make yourself sick with this type of vaccine -- over a week and then retest for antibodies?
If that still fails then consider figuring out how to perform your own mucus testing.
Replies from: TheSimplestExplanation, johnswentworth↑ comment by TheSimplestExplanation · 2021-02-04T23:13:28.695Z · LW(p) · GW(p)
Wasn't there something about to much antigen possibly reducing immune reaction in the white paper?
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-04T23:38:45.497Z · LW(p) · GW(p)
Yes, good job catching that. I'm currently using relatively low dose of each of nine different peptides, so there's a fair bit of elbow room before I hit a large dose of each individual peptide, and even more elbow room before I'd start worrying about tolerance. It is something I'll check if I start cranking up the dosage.
Also, to a large extent, "more dakka" here is as much about more time as higher dose. In general, you have to wait a while for a vaccine to really kick in.
↑ comment by johnswentworth · 2021-02-04T20:55:42.495Z · LW(p) · GW(p)
Yeah, more dakka is still the default plan right now if the next test comes back negative.
comment by dawangy · 2021-02-19T08:03:44.874Z · LW(p) · GW(p)
I asked Alex from RADVAC on reddit about antibody responses to the vaccine. He replied by saying 4 of the researchers saw a "positive antibody response."
https://www.reddit.com/r/radvac/comments/ig2b1q/related_news_open_for_suggestions/
"Some of our core group started performing ELISA assays to determine the presence of anti-Spike antibodies, beginning back in May/June. Since only a handful of us had at that point been collecting samples rigorously, the sample size available to us was small, so although we saw a positive antibody response, we didn't consider the (n=4) data credible as a data set."
I dare not interpret the implications of this finding on how much we should expect out of these vaccines, but I thought that this would be important to discuss.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-19T17:16:42.819Z · LW(p) · GW(p)
Ah excellent, thank you for getting ahold of someone. This matches the qualitative impression I had from the white paper.
It's not clear from that comment what the denominator is - i.e. 4 out of how many who tested for it? The white paper says that about 100 researchers had taken the vaccine (most of them presumably not as early as May/June), and the comment says "only a handful" were collecting samples rigorously as of May/June, so I'd guess ~10 or less. That gives ~40% chance or higher of antibody response with that version of the vaccine (where "or higher" includes a significant chance that only 4 people ran the ELISA assays, in which case Laplace' rule would give 80% chance of antibodies). Though note that the current version focuses on a different immunity strategy so it may not generalize, and the error bars were pretty wide to begin with.
One thing to note: if there's an antibody response in a significant fraction of people but not everyone, that's exactly the world where I'd expect "more dakka" to work.
comment by ChristianKl · 2021-02-06T13:04:47.496Z · LW(p) · GW(p)
From my reading it seems like people sneezing after applying nasal vaccines is one of the main error conditions. How about using a nose clamp after spraying the vaccine into the nose to prevent sneezing it out?
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-06T16:41:30.624Z · LW(p) · GW(p)
YES. Man, the most annoying part of this is you spray the stuff, and then have to spend an hour+ with your nose feeling runny but not blowing it.
comment by u8k · 2021-02-04T05:32:58.379Z · LW(p) · GW(p)
My jaw dropped at "~500 doses". At $2 per dose... that's significantly cheaper than it's costing the companies to produce the official vaccine. What am I missing here? I know you're making a different thing than Moderna, but if the thing your making exists and is this cheap then why is Pharma leaving the money on the floor and not mass producing this?
I think you're underselling the potential benefit here! Doing a batch of this and distributing it to 500 people may be unfeasible. I assume the FDA or something comes for you if you're trying to commercially distribute home-brewed vaccines.
But say, to 20 friends? Why not. Which brings the price down to $50 per/person, which seems like it'd be totally worth it to have immunity even a month earlier than the official vaccine is available to me. And it potentially gives immunity to your whole social scene.
I'll wait for your results, and then will strongly consider doing a batch of this myself.
Replies from: Dentin, Silver_Swift, johnswentworth, JacobKopczynski, Andrew_Clough↑ comment by Dentin · 2021-02-04T11:42:40.182Z · LW(p) · GW(p)
Oh, it's far, far worse (better?) than $2 per dose. As a thought experiment, I price estimated buying enough peptide for a hundred thousand doses, and it only costs about ten thousand dollars. Ten cents a dose is closer to realistic if you buy in bulk.
Which also brings to mind a question of civilizational inadequacy: if we really cared and it really mattered, why not have every university with a lab in the country crank out a hundred thousand doses per week to their local populace?
Replies from: ryan_b↑ comment by ryan_b · 2021-02-04T17:56:33.990Z · LW(p) · GW(p)
I don't think this has ever been possible in previous epidemics, at least for at-home manufacture.
That being said, I would strongly support compiling a list of low-risk interventions like this, so next time we can publicize them at the outset and try mitigating the problem while waiting for the low-risk&high-success solution to be developed.
↑ comment by Silver_Swift · 2021-02-04T12:07:30.598Z · LW(p) · GW(p)
If the thing your making exists and is this cheap then why is Pharma leaving the money on the floor and not mass producing this?
There are a number of costs that Moderna/Pfizer/Astrazenica incur that a homebrew vaccine does not. Of the top of my head:
1. Salaries for the (presumably highly educated) lab techs that put this stuff together. I don't know johnswentwort background, but presumably he wouldn't exactly be asking minimum wage if he was doing this commercially.
2. Costs of running large scale trials and going through all the paperwork to get FDA approval. I think I'm generally more in favour of organisations like the FDA than a lot of people
here, but even I expect this to be a very non-insignificant number.
3. Various taxes and costs of shipping/storing the vaccine until it can get to customers.
4. Costs of liability and a desire for the company to make a profit on this (as well as to pay the salaries for the all of the people needed to keep a large company running).
Given all that I don't think the gap between this and the commercial vaccines is that insane.
Replies from: Bucky↑ comment by Bucky · 2021-02-05T23:32:42.024Z · LW(p) · GW(p)
Trying to quantify these effects:
1. Salaries can't add much, especially if you're looking at mass producing. If you're creating 500 vaccines then maybe it takes a couple of hours? Say $20/hour (looking at local job listings for this kind of role) we get 8c/dose on salary. As you scale this is only going to go down.
2. It seems like vaccine trials can be done for a few hundred million although there is a big variation and I'm not completely sure whether the numbers given there include some manufacturing build up. If a large pharma company is going to be making lots of vaccine it seems like they should be able to achieve that for less than $1/dose.
3a. Taxes may add a decent few percent but can't be a main driver of cost
3b. Shipping costs for refrigerated goods are maybe 5c per 1000 miles per kg. That data is from a while back (1988!) and costs might be a bit higher for colder temperatures but I can't see this being a large fraction of the cost.
4a. For liability I note that at least AstraZeneca have struck deals in most countries to be exempt from such liabilities. It seems that in the US all COVID vaccines will benefit from this.
4b. Some companies (at least AstraZeneca and Johnson & Johnson) have said that they will be selling their COVID vaccines at cost. Even lacking this, I wouldn't expect corporate profits to be huge, even just from a PR point of view.
Additionally:
5. Risk of failed vaccine trials. If you only expect to have a 1 in 3 chance [LW(p) · GW(p)]of successful stage 3 trial then the $1/dose from 2 becomes $3/dose to expect to break even. I'm not sure whether this risk is covered by governments - I think it was to some extent but am not confident.
Given Dentin's comment that the material cost if something like 10c/dose (which makes sense given how little it cost to double John's peptides order) then I think most of the cost looks like it is in the trials and risk of failure thereof but this isn't enough to explain why companies aren't doing this. Its probably too late now anyway as vaccines already approved should have the pandemic under control before any new trials would be complete.
↑ comment by johnswentworth · 2021-02-04T17:31:40.972Z · LW(p) · GW(p)
why is Pharma leaving the money on the floor and not mass producing this?
Both the other comments here are on-point. The materials for large amounts of radvac would cost pennies per dose, because the cost-per-unit of the peptides drops very rapidly as you scale up (remember, we doubled our order for <10% extra cost).
However, in general, people and logistics are a bigger expense than materials for most products these days, and I certainly expect that to apply to vaccines.
↑ comment by Czynski (JacobKopczynski) · 2021-02-05T19:55:46.713Z · LW(p) · GW(p)
As someone else said, the logistical difficulties for this are much larger than the standard jab method, the production process doesn't scale well, and once prepared it doesn't last long even if you freeze it. Making this for 20 friends and distributing it to them is very plausibly net-harmful, because you need to gather in close quarters to hand off the prepared vaccine, do it within a fairly short window (1-3 days) for all 20, and then repeat all that, including the production of new doses, at least once per week and ideally 2-3x per week. Given that the only evidence for efficacy is in silico, none in vitro let alone in vivo or in actual humans, it's not at all clear that the benefits outweigh the increased risk of spread from all that close contact.
Replies from: kjz, ChristianKl, TheSimplestExplanation↑ comment by kjz · 2021-02-06T00:10:24.789Z · LW(p) · GW(p)
Why would they have to gather in close quarters? One person could make it in their kitchen, then leave the room while others come in one at a time to self administer their dose.
Replies from: JacobKopczynski↑ comment by Czynski (JacobKopczynski) · 2021-02-06T03:13:11.244Z · LW(p) · GW(p)
20 people sequentially, over a day or two, navigating an unfamiliar kitchen, without contact with the host? Not gonna happen. Most of them, at least, are going to have substantial exposure to the host (and vice versa).
Replies from: handoflixue↑ comment by handoflixue · 2021-02-06T11:04:08.879Z · LW(p) · GW(p)
It should not take more than 5 minutes to go in to the room, sit at the one available seat, locate the object placed on a bright red background, and use said inhaler. You open the window and run a fan, so that there is air circulation. If multiple people arrive at once, use cellphones to coordinate who goes in first - the other person sits in their car.
It really isn't challenging to make this safe, given the audience is "the sort of people who read LessWrong."
Replies from: kjz, JacobKopczynski↑ comment by kjz · 2021-02-06T17:20:53.361Z · LW(p) · GW(p)
Totally agree, and this is pretty much what I had in mind as well. The organizer can also host a Zoom call beforehand where they explain the procedure, answer any questions, and let people sign up for times spaced out by 5-10 minutes to self administer.
↑ comment by Czynski (JacobKopczynski) · 2021-02-19T05:59:05.342Z · LW(p) · GW(p)
None of that sounds like a thing most people attempting to arrange this will manage to do without exhausting some scarce resources; primarily willpower but also social capital, relationship closeness, and other fuzzy things. People on LessWrong are worse, not better, than the general population, both at weighing those costs and at bearing them.
↑ comment by ChristianKl · 2021-02-06T12:11:28.097Z · LW(p) · GW(p)
You don't need to gather in close quarters. You can simply drop it off in front of people's front doors and coordinate with mobile phones.
One person does the work of making the doses. Every other person gets tasked with doing one distribution run that they can either do themselves if they have a car/bike/motorcycle or outsource to another person (maybe a taskrabbit). They then travel around and put the microcentrifuge tubes either into postage box of someone or in front of their door when they are there and have the person open the door a minute later to pick up the microcentrifuge tube.
↑ comment by TheSimplestExplanation · 2021-02-08T10:38:55.384Z · LW(p) · GW(p)
Besides the distribution methods already proposed, one could depending on the circumstances, simply distribute it outside with masks.
↑ comment by Andrew_Clough · 2021-02-05T20:51:28.410Z · LW(p) · GW(p)
That's only for raw materials. Actual production seems to have involved a fair amount of skilled labor per dose which probably dominates the $2 figure. A commercial vaccine can automate that but then you've got lots of development costs as well as logistics and compliance to worry about. I wouldn't expect that this vaccine is especially cheap.
comment by Drake Morrison (Leviad) · 2022-12-10T21:56:19.837Z · LW(p) · GW(p)
A great example of taking the initiative and actually trying something that looks useful, even when it would be weird or frowned upon in normal society. I would like to see a post-review, but I'm not even sure if that matters. Going ahead and trying something that seems obviously useful, but weird and no one else is doing is already hard enough. This post was inspiring.
comment by johnswentworth · 2023-01-08T21:32:41.220Z · LW(p) · GW(p)
I did eventually get covid.
As was the general pattern with this whole RADVAC episode, it's ambiguous how much the vaccine helped. I caught covid in May 2022, about 15 months after the radvac doses, and about 13 months after my traditional vaccine (one shot J&J). In the intervening 15 months, my general mentality about covid was "I no longer need to give a shit"; I ate in restaurants multiple times per week, rode BART to and from the office daily, went to crowded places sometimes, traveled, and generally avoided wearing a mask insofar as that was socially acceptable.
comment by joetessier · 2021-02-20T09:36:08.658Z · LW(p) · GW(p)
This is a very interesting article and as long as the substances are very pure, it's probably low risk of any real health danger. However, I would really want to be very sure about that point before putting anything into my nasal passages.
As a scientist working in the pharmaceutical industry for many decades, I can say there are several big issues with this vaccine method. First, you will never know if it really works until you run blinded clinical trials against a placebo. This is the only way to tell and that is why it's required for any new drug/vaccine to be launched on the market. You can't just take a antibody test and see if it works. Even if there were the right antibody tests for these peptides, it might indicate some activity, but without using rigorous scientific method, there could be many other factors why you could see a response. Like you were exposed already to the virus and didn't know it.
Second immune response to peptides is significantly less effective generally. Whole viral proteins in the marketed vaccines are the best method to create an immune response. That is why almost all vaccines use some whole enzyme method or multiple proteins.
Finally the real problem with this method is that peptides are extremely unstable and very difficult to delivery into the body. That is why the most vaccines have to be injected, the oral and nasal dosage forms are specialized formulations.
Peptide memetics is a large field of study at both academic and pharmaceutical company labs and has been going on for many decades. Using peptides as drugs shows a huge amount of promise since is mimics the natural biological processes and are inherently effective, safe and non-toxic.
The problem has always been that it's extremely difficult to deliver peptides into the body. The bodies chemistry has a very large number of pathways to break down peptides and proteins. It is basic to biologic chemistry since almost all biologic processes are done by enzymes and peptides, and the body needs ways to break these down when they are finished their job. This delivery issue is why most peptide drugs today are in an injectable dosage form. It's the only good way to get them into the blood steam and have them circulating through the body. They still quickly breakdown especially after entering the cells, but usually last long enough to have their intended affect. I can tell you that there have been a very large number of peptide drugs that have shown real promise, but this stability issue has limited their application and the reason that there are still so few on the market even after decades of research. We have developed tricks in the industry, the main one being to change the peptide structure to make them more stable and those can work, but then the activity can be substantially lower. It is a very difficult problem that continues to be studied.
Today there are several nasal dosage forms of peptide drugs on the market, however these usually have very specialized formulations, that include stabilizers, special particle size ranges and shapes that enhance absorption or even nanoparticles. However as noted above, the first cells they see in the nasal passage will start to digest them quickly. I doubt very much if any of the peptide formulated up in your kitchen will actually make it into the blood stream where it could then effect the immunity.
I won't say don't try it, but there are always a serious risks and most likely your just wasting time and money. Hey, it's surely better then injecting bleach!
I'd be happy to discuss with anyone if they would like more information.
Replies from: kjz↑ comment by kjz · 2021-02-20T13:47:35.226Z · LW(p) · GW(p)
Welcome, and thanks for making your first comment!
As a fellow scientist with decades of experience in the industry, I disagree with several of your claims.
First, you will never know if it really works until you run blinded clinical trials against a placebo. This is the only way to tell and that is why it's required for any new drug/vaccine to be launched on the market.
Clinical trials are helpful for understanding whether a drug/vaccine works on the population level. But on the individual level, clinical trials are not the only way to tell. For example, you can just take an antibody test and see if it works.
You can't just take a antibody test and see if it works.
Of course you can.
Even if there were the right antibody tests for these peptides
Anna Czarnota posted an initial protocol here [LW(p) · GW(p)]. I haven't tried it, but it seems reasonable and likely to provide useful information about one's level of protection.
but without using rigorous scientific method, there could be many other factors why you could see a response. Like you were exposed already to the virus and didn't know it.
The "rigorous scientific method" is not the only way to generate knowledge that allows individuals to update their priors. But setting that aside, the question of whether one's immune response came from the vaccine or from previous exposure to the virus is not very relevant to one's future decision making. Either way, the antibody test provides information about one's current level of immunity, which one can use to update their risk tolerance and behaviors.
It feels like your comments are aimed at the question, "What is the best vaccine (or vaccines) to approve and mass produce for the general population?" which is a perfectly valid and important question. As things currently stand, this relies on the standard clinical trials/FDA approval process. But this process takes a long time and is prone to all sorts of delays and inefficiencies due to politics and organizational maze behaviors, during which the pandemic continues to spread. Realizing that, the radvac developers and many commenters here have been asking a different question: "What can individuals do now (or in a future pandemic) to mitigate their personal risk of being infected?"
Both questions are important, but the large organizations responsible for developing/approving new vaccines have very different incentives than individuals looking for ways to minimize their own risk of infection.
Replies from: joetessier↑ comment by joetessier · 2021-02-21T09:17:40.827Z · LW(p) · GW(p)
As you can tell, I'm have more of a mainstream approach to health issues. You are totally correct that delays and avoidance of risk in the development process of the pharma industry and with the government clauses personal suffering every day. And let's not even start about the the pharmaceutical industry's need for "commercial success" before they even start working on a disease or problem.
However, I think you may have missed my main premise, the fact that degradation is a huge hurtle in any peptide application, it is a clear fact that peptides are digested extremely quickly and therefore are very ineffective as any type of drug. You are correct that the antibody test presented could at least show if these peptides actually enter the body and if you are getting an immune response to these specific peptides. I would be very interested to hear if anyone has tried it on themselves and gotten positive results. However, the other problem here is that even if your body does develop a immune response to peptides of the virus, it does not necessarily correlate to having a immune response to the virus itself.
I can see a pathway for this method to develop immunity to the virus itself, where the virus enzymes are broken down to the peptides you have taken and have a general immunity response offering some protection. However as a scientist personally I would need to see proof before trying this method. That would start with some lab trials showing these antibodies have and affect. Then of course we are back to clinical trials which are really the only way to prove this method work. And as you note this takes a lot of time while people are suffering and dying.
So my final point is sure, you can try this method while you are waiting for the vaccine, but it definitely shouldn't be a replacement for the vaccine. Also you should keep in mind there are still some risks of something untested and mixed up in your kitchen, but hey, I'm not stopping anyone.
Finally if you really want to protect yourself from the virus until you can get the vaccine, then I suggest you follow the swiss cheese advise.
The best way to have some level of protection is to follow the recommendations of the epidemiologists and be extremely careful about your behavior. I know a lot of people can't do this due to work and personal reasons, but honestly if everyone were just more careful about how they act in this situation, we all would be better off.
Stay safe out there!
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-21T11:28:06.661Z · LW(p) · GW(p)
The best way to have some level of protection is to follow the recommendations of the epidemiologists and be extremely careful about your behavior.
I would be doubtful that the best way is to listening to someone who hasn't on their list avoiding closed spaces, ventilation and humidity (in closed spaces), avoiding touching common buttons with fingers and recommends cloth masks (as opposed to reused FFP2/3) but has cleaning/desinfection on their list a good source of information.
Then of course we are back to clinical trials which are really the only way to prove this method work.
Then why do you link to advice that recommends unproven interventions like desinfection and using gloves when serving food?
comment by TheSimplestExplanation · 2021-02-04T13:30:30.106Z · LW(p) · GW(p)
From the white paper:
Antibody can be measured in nasal wash, however this is less quantitative and reliable than measurement in serum.
comment by Anna Czarnota (anna-czarnota) · 2021-02-10T11:41:51.886Z · LW(p) · GW(p)
Hi,
It's quite interesting what you are doing. Have you thought about testing immune response yourself using peptides from the vaccine? Of course you can only run humoral responses, but it would give you far more accurate results then any commercially available test. You could bind peptides to the high-binding plates, use your mucosa samples and detect with universal anti-human IgM+IgG+IgG antibodies conjugated with HRP.
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-10T11:59:43.300Z · LW(p) · GW(p)
This sounds to me like the kind of thing biology expertise to get right and potentially more equipment.
Replies from: anna-czarnota↑ comment by Anna Czarnota (anna-czarnota) · 2021-02-10T12:53:10.007Z · LW(p) · GW(p)
This may be true, I'm talking about ELISA test. In theory you only need high binding plates, secondary antibodies and some kind of detection (TMB + H2SO4 give colorimetric effect and you can try and skip plate reader). I understand that it can be quite complicated to perform, but I am wondering how you plan to chceck the immune response against Orf1 and Orf 8. You could check blood against Spike and nucleocapside (you need to check if antigen test is using full length Spike and Nuc), but due to the use of peptides, lenghth of the peptides and expected low IgG titer, detection (as you said) can get tricky. But congrats anyway.
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-10T14:10:14.439Z · LW(p) · GW(p)
This may be true, I'm talking about ELISA test.
Back in university I did a ELISA test. It's a bit fuzzy in my memory but I think we did a lot to purify the solution we were working with before we did ELISA. Do you think the concentrations would here be high enough that you can just do ELISA and see something with the naked eye?
Replies from: anna-czarnota↑ comment by Anna Czarnota (anna-czarnota) · 2021-02-10T14:55:40.717Z · LW(p) · GW(p)
I think that you can perform ELISA using unpurified nasal wash or diluted mucus. I often perform ELISA using sera/full blood or unpurified cell lysates. I would do a nasal or throat swab and put it in phosphate buffer (or PBS). You can further dilute samples if the viscosity is too high. ELISA is very sensitive and my guess is, you should be able to see positive signal without plate reader. It very much depends on both antigen used in ELISA and antibody concentration. For peptides such as yours (without tags) I use concentration of about 10-20 ug/mL, sera diluted 1:1000-1:10000 and higher and still get good, clear signal. Of course you don't want to dilute your nasal samples so much, but undiluted, 1:5 or 1:20 could give nice reading, hopefully without any background. EDIT: I know it may sound complicated, but I just wanted you to know that checking immune response against antigen used (in this case peptides) is a possibility.
Replies from: johnswentworth, kjz, ChristianKl↑ comment by johnswentworth · 2021-02-10T23:28:02.724Z · LW(p) · GW(p)
This was a very useful thread. Thankyou and strong upvote.
↑ comment by kjz · 2021-02-10T23:15:12.574Z · LW(p) · GW(p)
Is this something that can be done at home with readily available and affordable equipment? If so, would you be willing to share more details of how someone might get started? I think a lot of readers would be interested in hearing more about this - it could even be its own post.
Replies from: anna-czarnota, johnswentworth↑ comment by Anna Czarnota (anna-czarnota) · 2021-02-12T13:09:16.185Z · LW(p) · GW(p)
Hi,
I wrote down this manual for anyone to follow - https://drive.google.com/file/d/1qtfyE42ECZAqSFpqGR9JawX3YoVAvVSg/view?usp=sharing
As it is a modified protocol I'm not sure It will work (especially when we do not have any positive and negative baseline).
In short:
Antigen (peptide) binding to the plates:
- Prepare peptide solutions in PBS buffer in 1.5 mL tubes. The final concentration of the peptides should be around 10-20 µg/mL
- Prepare high-binding 96-well plates and a piece of tin foil for cover. Add a 100 µL of peptide solution to each experimental well. Cover the plate and incubate at 4oC over night (16h).
- Take the plate from the fridge and discard peptide solution
- Wash the plate by adding wash buffer (approx. 200 µL per well).
Blocking:
- Add approx. 200 µL of blocking solution to each well. Incubate for 2h at room temperature and discard.
- Wash the plate 3 times as described above.
Addition of the primary antibodies (from nasal swabs):
- Prepare the nasal swab/wash samples. Collected swabs should be stored in PBS. Nasal wash can be performed using PBS. The samples can be applied to the wells without dilution, but I would suggest further dilution of the samples using antibody buffer.
- Add prepared nasal samples – 100 µL/well. Incubate at 4oC overnight.
- Discard samples and wash the plate 4 times as described above.
Addition of secondary antibodies conjugated with HRP:
- Prepare the solution of secondary antibodies (1:2500) in antibody buffer.
- Add secondary antibodies to the plate – 100 µL/well. Incubate for 90 min. at room temperature.
- Discard secondary antibodies and wash 5 times as described above.
- Add 100 µL of TMB to each well. The reaction should turn the solution blue. The time of the incubation can vary from 1 min. to 30 minutes.
- If you want you can stop the reaction by adding 50 µL of 0.5 M H2SO4 – this will turn solution yellow.
I guess anything with a signal stronger then control can be considered positive signal. Usually for my experiments I'm using the cutoff of "three times value of the blank or more (after subtraction of the proper negative control)". If you do not have negative control you can only use blank as a point of reference.
Replies from: johnswentworth, jacobjacob, kjz↑ comment by johnswentworth · 2021-02-12T17:28:44.876Z · LW(p) · GW(p)
You are officially The Best.
↑ comment by Bird Concept (jacobjacob) · 2021-02-23T00:52:35.702Z · LW(p) · GW(p)
Curious if anyone ended up running this process, and, if so, what your results were?
↑ comment by johnswentworth · 2021-02-11T00:03:16.240Z · LW(p) · GW(p)
I'd recommend googling for "ELISA kit", and reading up on exactly how it works. My understanding is that it shouldn't require particularly fancy equipment as long as the sample prep is simple (in particular, no microcentrifuge) and the signal is strong enough to read with the naked eye. If unpurified nasal wash/diluted mucus works and the signal is strong (as Anna suggests), then it should be viable.
There is a fair bit of complexity, but it's the kind of complexity that involves lots of straightforward steps rather than anything confusing/difficult. Anna's comments make me a lot more optimistic that it's viable without any expensive equipment.
↑ comment by ChristianKl · 2021-02-10T16:07:20.658Z · LW(p) · GW(p)
Thanks for explaning it. Maybe someone here will put it into practice.
comment by cursed · 2021-02-04T06:39:26.224Z · LW(p) · GW(p)
Props to you for taking action here, this is some impressive stuff.
That being said, I'm extremely skeptical that this will work, my belief is that there's a 1-2% chance here that you've effectively immunized yourself from COVID.
What do you believe is the probability of success?
Why are established pharmaceutical companies spending billions on research and using complex mRNA vaccines when simply creating some peptides and adding it to a solution works just as well?
Replies from: Dentin↑ comment by Dentin · 2021-02-04T12:07:44.928Z · LW(p) · GW(p)
My rough guess is that there's a 75% probability of effectively full immunity, and a 90% probability of severity reduction. This is a pretty well tested and understood vaccine mechanism, and the goal isn't "perfect immunity" as "prime the immune system so it doesn't spend a week guessing about what antibodies it needs to combat the virus effectively".
As to why established companies don't do it, I believe it's partially logistics, and largely red tape. Logisitics first (though it should be noted that at least some of these could likely be tackled with a bit of effort):
- Shots are well understood and easy; people are used to them, people know how to give them, etc. Nasal spray is irritating and makes you want to blow your nose, which washes out a lot of it and reduces effectiveness.
- You need multiple of these annoying doses in the nose, staggered a few days apart, to generate a 'good' response.
- This particular nanoparticle vaccine doesn't have a long shelf life due to peptide degradation. Peptides don't last forever, and while they're more stable than the RNA vaccines, you'd have to ship them frozen as well.
- Nanoparticle vaccines in general suffer from particle aggregation over time. The particles will gradually aggregate in solution, and if you freeze it they aggregate faster; freezing changes the size distribution pretty dramatically. That said, I don't know how much that impacts effectiveness, and it doesn't seem like an extensively researched topic. I only found one paper discussing it.
- Because it doesn't have a long shelf life, it has to be mixed in-house, then distributed, preferably within a small number of days. I plan to mix a new batch every week for my prime and boosters. On the plus side, mixing it can actually be done at home with pretty cheap and ordinary tools, it just takes time.
Red tape on the other hand is a huge problem and flat out intractable. Unlike injections, companies need to get safety approvals and testing done for pretty much all the components and the delivery mechanism, not just the active ingredients (the peptides.) Then, separate approvals and testing for each individual set of peptides, via a process that operates closer to the scale of decades than months and costs billions of dollars. Any modification to the peptide set can be expected to restart the process from scratch. Then, companies also need to set up / reconfigure their legal strategy to protect themselves, and ensure that things are sufficiently balanced business-wise that they don't go bankrupt.
Using ancient, primitive "stab people in the arm" technologies isn't great, but it erases likely more than half of the regulatory burden, because "we've been doing that for a century and we know it's only somewhat dangerous", wheras new technologies like "snort some nanoparticles" are Scary and Dangerous and New and Have Not Yet Been Approved By Appropriately Serious People Being Serious.
Nevermind that every time you play with a pet, breathe in part of a cloud of dust, or smell your SO's hair, you're inhaling more foreign peptides than what's present in a vaccine dose.
Replies from: dxu, cursed↑ comment by dxu · 2021-02-04T20:24:16.344Z · LW(p) · GW(p)
This is a very in-depth explanation of some of the constraints affecting pharmaceutical companies that (mostly) don't apply to individuals, and is useful as an object-level explanation for those interested. I'm glad this comment was written, and I upvoted accordingly.
Having said that, I would also like to point out that a detailed explanation of the constraints shouldn't be needed to address the argument in the grandparent comment, which simply reads:
Why are established pharmaceutical companies spending billions on research and using complex mRNA vaccines when simply creating some peptides and adding it to a solution works just as well?
This question inherently assumes that the situation with commercial vaccine-makers is efficient with respect to easy, do-it-yourself interventions, and the key point I want to make is that this assumption is unjustified even if you don't happen to have access to a handy list of bullet points detailing the ways in which companies and individuals differ on this front. (Eliezer wrote a whole book on this at one point, from which I'll quote a relevant section:)
My wife has a severe case of Seasonal Affective Disorder. As of 2014, she’d tried sitting in front of a little lightbox for an hour per day, and it hadn’t worked. SAD’s effects were crippling enough for it to be worth our time to consider extreme options, like her spending time in South America during the winter months. And indeed, vacationing in Chile and receiving more exposure to actual sunlight did work, where lightboxes failed.
From my perspective, the obvious next thought was: “Empirically, dinky little lightboxes don’t work. Empirically, the Sun does work. Next step: more light. Fill our house with more lumens than lightboxes provide.” In short order, I had strung up sixty-five 60W-equivalent LED bulbs in the living room, and another sixty-five in her bedroom.
Ah, but should I assume that my civilization is being opportunistic about seeking out ways to cure SAD, and that if putting up 130 LED light bulbs often worked when lightboxes failed, doctors would already know about that? Should the fact that putting up 130 light bulbs isn’t a well-known next step after lightboxes convince me that my bright idea is probably not a good idea, because if it were, everyone would already be doing it? Should I conclude from my inability to find any published studies on the Internet testing this question that there is some fatal flaw in my plan that I’m just not seeing?
We might call this argument “Chesterton’s Absence of a Fence.” The thought being: I shouldn’t build a fence here, because if it were a good idea to have a fence here, someone would already have built it. The underlying question here is: How strongly should I expect that this extremely common medical problem has been thoroughly considered by my civilization, and that there’s nothing new, effective, and unconventional that I can personally improvise?
Eyeballing this question, my off-the-cuff answer—based mostly on the impressions related to me by every friend of mine who has ever dealt with medicine on a research level—is that I wouldn’t necessarily expect any medical researcher ever to have done a formal experiment on the first thought that popped into my mind for treating this extremely common depressive syndrome. Nor would I strongly expect the intervention, if initial tests found it to be effective, to have received enough attention that I could Google it.
The grandparent comment is more or less an exact example of this species of argument, and is the first of its kind that I can recall seeing "in the wild". I think examples of this kind of thinking are all over the place, but it's rare to find a case where somebody explicitly deploys an argument of this type in such a direct, obvious way. So I wanted to draw attention to this, with further emphasis on the idea that such arguments are not valid in general.
The prevalence of this kind of thinking is why (I claim) at-home, do-it-yourself interventions are so uncommon, and why this particular intervention went largely unnoticed even among the rationalist community. It's a failure mode that's easy to slip into, so I think it's important to point these things out explicitly and push back against them when they're spotted (which is the reason I wrote this comment).
IMPORTANT NOTE: This should be obvious enough to anyone who read Inadequate Equilibria, but one thing I'm not saying here is that you should just trust random advice you find online. You should obviously perform an object-level evaluation of the advice, and put substantial effort into investigating potential risks; such an assessment might very well require multiple days' or weeks' worth of work, and end up including such things as the bulleted list in the parent comment. The point is that once you've performed that assessment, it serves no further purpose to question yourself based only on the fact that others aren't doing the thing you're doing; this is what Eliezer would call wasted motion [LW · GW], and it's unproductive at best and harmful at worst. If you find yourself thinking along these lines, you should stop, in particular if you find yourself saying things like this (emphasis mine):
That being said, I'm extremely skeptical that this will work, my belief is that there's a 1-2% chance here that you've effectively immunized yourself from COVID.
You cannot get enough Bayesian evidence from the fact that [insert company here] isn't doing [insert intervention here] to reduce your probability of an intervention being effective all the way down to 1-2%. That 1-2% figure almost certainly didn't come from any attempt at a numerical assessment; rather, it came purely from an abstract intuition that "stuff that isn't officially endorsed doesn't work". This is the kind of thinking that (I assert) should be noticed and stamped out.
Replies from: Dentin, cursed↑ comment by Dentin · 2021-02-04T23:31:04.073Z · LW(p) · GW(p)
While I generally agree with the concept, I'm going to push back a little here. I read the 1-2% chance as less being about "why aren't companies doing it" and more about lack of information.
My initial reaction to seeing it was that it was a combination factors along the lines of:
- "there's a lot of fraud out there, and by default my prior for things like this being valid is very low"
- "factoring in that a couple of lesswrongers seem to think it's ok that only pushes my estimate up into the handful of percent range"
- "but there's also evidence against, in that we don't see any commercial products based on this, which pushes my estimate down to 1-2%"
I think this is a pretty reasonable place to start from.
↑ comment by cursed · 2021-02-05T01:56:11.178Z · LW(p) · GW(p)
I don't understand the argument about SAD.
Should I conclude from my inability to find any published studies on the Internet testing this question that there is some fatal flaw in my plan that I’m just not seeing?
A simple Google search shows thousands of articles addressing this very solution. The first Google result I found is a paper from 1984 with 2,758 citations: https://jamanetwork.com/journals/jamapsychiatry/article-abstract/493246
We report our preliminary attempts to modify these depressions by manipulating environmental lighting conditions. We have recently reported reversing depression in one patient with SAD by modifying his environmental lighting
...
The following light treatment was administered. ... (1) bright, white full-spectrum fluorescent light (approximately 2,500 lux at 90 cm)
But taking a step back, the "Chesterton’s Absence of a Fence" argument doesn't apply here because the circumstances are very different. The entire world is desperately looking for a way to stop COVID. If SAD suddenly occurred out of nowhere and affected the entire economy, you would be sure that bright lights would be one of the first things to be tested.
Dentin addresses the 1-2% claim pretty well, so I won't repeat it.
Replies from: dxu↑ comment by dxu · 2021-02-05T16:37:56.012Z · LW(p) · GW(p)
A simple Google search shows thousands of articles addressing this very solution.
The solution in the paper you link is literally the solution Eliezer described trying, and not working:
As of 2014, she’d tried sitting in front of a little lightbox for an hour per day, and it hadn’t worked.
(Note that the "little lightbox" in question was very likely one of these, which you may notice have mostly ratings of 10,000 lux rather than the 2,500 cited in the paper. So, significantly brighter, and despite that, didn't work.)
It does sound like you misunderstood, in other words. Knowing that light exposure is an effective treatment for SAD is indeed a known solution; this is why Eliezer tried light boxes to begin with. The point of that excerpt is that this "known solution" did not work for his wife, and the obvious next step of scaling up the amount of light used was not investigated in any of the clinical literature.
But taking a step back, the "Chesterton’s Absence of a Fence" argument doesn't apply here because the circumstances are very different. The entire world is desperately looking for a way to stop COVID. If SAD suddenly occurred out of nowhere and affected the entire economy, you would be sure that bright lights would be one of the first things to be tested.
This is simply a (slightly) disguised variation of your original argument. Absent strong reasons to expect to see efficiency, you should not expect to see efficiency. The "entire world desperately looking for a way to stop COVID" led to bungled vaccine distribution, delayed production, supply shortages, the list goes on and on. Empirically, we do not observe anything close to efficiency in this market, and this should be obvious even without the aid of Dentin's list of bullet points (though naturally those bullet points are very helpful).
(Question: did seeing those bullet points cause you to update at all in the direction of this working, or are you sticking with your 1-2% prior? The latter seems fairly indefensible from an epistemic standpoint, I think.)
Not only is the argument above flawed, it's also special pleading with respect to COVID. Here is the analogue of your argument with respect to SAD:
Around 7% of the population has severe Seasonal Affective Disorder, and another 20% or so has weak Seasonal Affective Disorder. Around 50% of tested cases respond to standard lightboxes. So if the intervention of stringing up a hundred LED bulbs actually worked, it could provide a major improvement to the lives of 3% of the US population, costing on the order of $1000 each (without economies of scale). Many of those 9 million US citizens would be rich enough to afford that as a treatment for major winter depression. If you could prove that your system worked, you could create a company to sell SAD-grade lighting systems and have a large market.
SAD is not an uncommon disorder. In terms of QALYs lost, it's... probably not directly comparable with COVID, but it's at the very least in the same ballpark--certainly to the point where "people want to stop COVID, but they don't care about SAD" is clearly false.
And yet, in point of fact, there are no papers describing the unspeakably obvious intervention of "if your lights don't seem to be working, use more lights", nor are there any companies predicated on this idea. If Eliezer had followed your reasoning to its end conclusion, he might not have bothered testing more light... except that his background assumptions did not imply the (again, fairly indefensible, in my view) heuristic that "if no one else is doing it, the only possible explanation is that it must not work, else people are forgoing free money". And as a result, he did try the intervention, and it worked, and (we can assume) his wife's quality of life was improved significantly as a result.
If there's an argument that (a) applies in full generality to anything other people haven't done before, and (b) if applied, would regularly lead people to forgo testing out their ideas (and not due to any object-level concerns, either, e.g. maybe it's a risky idea to test), then I assert that that argument is bad and harmful, and that you should stop reasoning in this manner.
↑ comment by cursed · 2021-02-05T01:40:27.496Z · LW(p) · GW(p)
You can buy nasal sprays over-the-counter, while I can't think of a single injectable medicine that you can buy legally without a prescription. I don't think the "stab people in the arm" argument is very strong.
Would you like to make a friendly wager? (Either Dentin, or johnswentworth, or anyone else making their own vaccine). We can do 50/50, since its in between our estimates. If you have two positive back-to-back anti-body tests within 2 months, you win (assuming you don't actually contract covid, which I trust you'll be honest here). If not, I win. To start off with, I'm willing to put down $100, but happy to go up or down.
Replies from: johnswentworth, Dentin↑ comment by johnswentworth · 2021-02-05T02:46:10.574Z · LW(p) · GW(p)
I wouldn't take 50/50. I do think it's much more likely than that to induce mucus antibodies, but not blood antibodies. I would take 3:1 odds.
↑ comment by Dentin · 2021-02-05T11:44:53.732Z · LW(p) · GW(p)
My estimate for whether or not I would test positive on a blood test was only about 50%, since blood isn't the primary place that the response is generated. I'm already betting a substantial amount of money (peptide purchases and equipment) that this will be helpful, and I see no reason to throw an additional $50 on a break-even bet here.
I would, however, be happy to commit to sharing results, whether they be positive or negative.
... and now it occurs to me that if Lesswrong had a 'public precommitments' feature, I would totally use it.
comment by Jsevillamol · 2021-02-10T12:55:37.856Z · LW(p) · GW(p)
Amazing initiative John - you might give yourself a D but I am giving you an A+ no doubt.
Trying to decide if I should recommend this to my family.
In Spain, we have 18000 confirmed COVID cases in January 2021. I assume real cases are at least 20000. Some projections estimate that laypeople might not get vaccinated in 10 months, so the potential benefit of a widespread DIY vaccine is avoiding 200k cases of COVID19 (optimistically assuming linear growth of cases).
Spain pop is 47 million, so the naïve chance of COVID for an individual before vaccines are widely available is 2e4*10 / 5e6 ie about 1 in 250.
Let's say that the DIY vaccine has 10% chance of working on a givne individual. If we take the side effects of the vaccine to be as bad as catching COVID19 itself, then I want the chances of a serious side effect to be lower than 1 in 2500 for the DIY vaccine to be worth it.
Taking into account the risk of preparing it incorrectly plus general precaution, the chances of a serious side effect look to me more like 1 in 100 than 1 in 1000.
So I do not think, given my beliefs, that I should recommend it. Is this reasoning broadly correct? What is a good baseline for the chances of a side effect in a new peptide vaccine?
↑ comment by johnswentworth · 2021-02-10T16:51:12.255Z · LW(p) · GW(p)
I think the chances of side effects as serious as COVID are much lower than that. I also think the chances of success are higher, but on that front it probably makes sense for you to wait a few weeks at least until I, and possibly others, have some test results.
But my biggest difference from this calculation is that I'd account for the benefits of not-having-to-try-hard-to-avoid-COVID. The chance of catching it is only low if you avoid going out and socializing; with a successful vaccine, you can have a low chance of catching it without paying that cost.
So that's how my calculation would differ. On a meta level:
So I do not think, given my beliefs, that I should recommend it. Is this reasoning broadly correct?
... your reasoning is reasonable, and I do not think anyone should defer to my reasoning on this subject; I do not have enough legible expertise to merit that. Upvote for writing out the calculation.
I do not know the baseline for chances of a side effect in a new peptide vaccine. I expect that if side effects anywhere near as bad as COVID happened with any non-negligible rate, then I would have heard about it already, but I don't have numbers or data on it.
comment by magfrump · 2021-02-07T04:51:46.612Z · LW(p) · GW(p)
How were you able to find someone who would give you an antibody test?
I made some effort to get an antibody test a few weeks ago but multiple sources refused to order or run one, even after I had an appointment that I showed up for in person.
Replies from: johnswentworth, fluffyclouds↑ comment by johnswentworth · 2021-02-07T04:58:44.233Z · LW(p) · GW(p)
Huh. I believe we ordered from the labcorp website, didn't have any problems.
↑ comment by fluffyclouds · 2021-02-07T20:13:05.219Z · LW(p) · GW(p)
Cerascreen offers an at-home antibody test. You use the kit you buy to draw a small blood sample at home and mail it to them. They use the ELISA method to test the blood for IgG antibodies and show you the result on a webpage. Not sure if this is available outside Germany, though maybe a different company offers something like this where you live.
Abbott also produces a blood test for IgG/IgM antibodies, except that it comes with a small test cassette that gives you the result directly, without sending it to a lab. Maybe importing this (or something similar) is an option for you.
comment by Brendan Long (korin43) · 2021-02-05T03:45:58.535Z · LW(p) · GW(p)
FYI, there's a map of people working on this that you can add yourself to for coordination:
https://radvac.org/researchers-map/
Replies from: Jayson_Virissimo↑ comment by Jayson_Virissimo · 2021-02-05T16:52:40.881Z · LW(p) · GW(p)
Does this add you to an email list where discussion is happening, or merely put you on a map so that others in the area can reach out to you on an ad hoc basis?
Replies from: korin43↑ comment by Brendan Long (korin43) · 2021-02-05T17:29:22.813Z · LW(p) · GW(p)
I think it just puts you on the map.
comment by Adele Lopez (adele-lopez-1) · 2021-02-04T23:03:45.825Z · LW(p) · GW(p)
The cheapest options suffice for the vaccine - the peptides don’t need to be “purified” (this just means removing partial sequences), they don’t need any special modifications, and very small amounts suffice.
Seems like partial sequences could contribute to the autoimmune disease risk since they would be much more likely to match to normal cellular proteins. Has this been considered?
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-04T23:34:33.454Z · LW(p) · GW(p)
Just based on general background knowledge, if autoimmune problems were induced by sequences short enough to show up frequently from random noise, then we'd have autoimmune problems all the time (since we're breathing in peptides - including partially-broken-down-peptides - basically every day). So I'd be very surprised if it were an issue.
This is exactly the sort of question I love to see, though. Great thinking.
Replies from: adele-lopez-1↑ comment by Adele Lopez (adele-lopez-1) · 2021-02-05T01:16:24.554Z · LW(p) · GW(p)
So at what point does a peptide go from triggering an immune reaction, to being completely inert?
Off the top of my head, I can think of three possible factors:
- The length of the peptide matters. Presumably, your body wouldn't create an immune reaction to dipeptides under any circumstances. But in this case, how close are these peptides to the threshold length? If they're well above it, then it seems this risk is still relevant. If they're close to the threshold, then that would mitigate a lot of this risk.
- The amount of the peptide matters. I'd expect that your immune system generally does not create an immune response to a single foreign peptide it detects, and that there's a certain amount (perhaps variable between different peptides) necessary to cause a reaction. That would explain why breathing doesn't create autoimmune issues. But in this case, we might still expect a particular partial peptide to have a significant population, especially since certain peptide-peptide bonds are weaker than others. So again, this risk is still relevant.
- The peptide must be associated with some sort of damage. It's plausible that the immune system doesn't create a response unless it detects actual damage (irritation at the human level). In that case, the vaccine presumably works because snorting a spray is sufficiently irritating. This risk is clearly still relevant in this case.
I suspect that the actual answer is likely a combination of all three, and maybe some other stuff is relevant.
Also, I checked the white paper, and it says the partial peptides aren't an issue, but doesn't seem to elaborate.
Replies from: adele-lopez-1↑ comment by Adele Lopez (adele-lopez-1) · 2021-02-05T01:51:35.537Z · LW(p) · GW(p)
Alright, so the thing that antibodies are "trained" to detect is called an epitope. On page 23 of the white paper, it says:
- VED might arise through vaccine design-induced distortion of viral epitopes, as has been proposed to occur by formalin treatment of RSV. a. Our preferred epitope type is synthetic peptides. Such peptides are chemically well defined and have a high degree of structural integrity.
So distortions of the epitope can cause an autoimmune reaction, and synthetic peptides are good because they're stable and you know exactly what you are getting. But if you have a bunch of partial peptides mixed in, then it substantially weakens this point.
Re: 3. It does seem to be true that "damage" is required. Substances which provoke an immune response are called adjuvants, and the adjuvants in this vaccine are the Chitosan and the Tripolyphosphate.
Reading the paper more closely, it says that the truncated peptides are fine because the peptides get chopped up by proteases anyway. This does make me feel a bit less worried about this, but it also implies that this would be a potential issue for purified peptides as well.
ETA: I also did a spot-check to see at which point this would actually become an issue with the actual peptides in the vaccine using https://db.systemsbiology.net/sbeams/cgi/PeptideAtlas/Search It seems you can generally expect length 6 sequences of peptides in radvac to be unique to the virus, while length 4 sequences of peptides from radvac are typically widespread.
comment by ChristianKl · 2021-02-04T18:38:51.826Z · LW(p) · GW(p)
Are there other nasal vaccines that are based on nanopeptides currently on the market and FDA-approved?
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-04T19:30:41.015Z · LW(p) · GW(p)
I don't know this, but the whitepaper does mention that this approach is quite common for research vaccines.
comment by benjaminikuta · 2021-02-04T01:21:55.960Z · LW(p) · GW(p)
How do you know this is safe?
Replies from: Dentin, johnswentworth↑ comment by Dentin · 2021-02-04T12:15:19.235Z · LW(p) · GW(p)
The whitepaper is a good source, but like johnswentworth, I also contacted a medical professional to evaluate it. The response came back quickly and confidently, and was along these lines:
"Oh, yeah, this is safe. Nasal vaccines are safe. The biggest worry is that it might not work, so make sure you get the commercial vaccine too. I'd be interested in doing this with you as a joint project and giving it to my family, and I also have a colleague who might be interested in doing it."
The biggest point of disbelief on their part was that it's possible to order all the equipment and peptides online and have them shipped to your door.
↑ comment by johnswentworth · 2021-02-04T02:23:41.395Z · LW(p) · GW(p)
See the answers to gwillen above, and also the whitepaper.
comment by cousin_it · 2021-02-06T00:43:12.440Z · LW(p) · GW(p)
By now, about 17 out of every 100k people aged 35-44 in the US have died from corona (source). Assuming this number doesn't grow a lot, "let's try an amateur medical thing" seems more risky, if only due to unknown unknowns. Am I missing something?
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-08T17:41:25.024Z · LW(p) · GW(p)
Two things. First, death isn't the only downside of coronavirus. Even losing your sense of smell for six months sucks. (My mother caught COVID in March, and spent the next few months complaining that her food tasted like dirt.) And of course there's other forms of damage as well.
Second, and more importantly, control systems. The death number (in any age band) is low in large part because people are staying home and not socializing. The chance of death I would create for myself and those around me by going out and/or socializing is probably higher than 17/100k. The upside of immunity (in particular measurable immunity) is that I can increase my exposure without breaking the control system.
Replies from: cousin_itcomment by koroviev · 2021-02-08T01:10:34.673Z · LW(p) · GW(p)
Thank you for both conducting this experiment as well as for writing it up in great detail.
How much study do you estimate someone would have to invest into biology (or biohacking) to at least understand what's going on here? What are some "genres" of biology someone could start looking into to understand this work?
This looks like a really interesting area to probe, but my current level of knowledge in biology ("1 year of high school biology classes") makes it difficult to even formulate questions or google queries about it.
Replies from: Dentin, johnswentworth↑ comment by Dentin · 2021-02-10T00:18:40.545Z · LW(p) · GW(p)
In my case, I'd estimate that I've spent around two hundred hours over the last several months coming sufficiently up to speed on the topics that I can reason about them. I started with about your level of biology (or possibly less), but probably a slightly stronger chemistry background.
For the basics, I started with cell biochemistry, DNA/RNA, mRNA and protein construction. From the vaccine side of things, I just started looking up things I found in the whitepaper which I didn't understand, and once I understood all the terms I started looking for and reading research papers. When I found something I wasn't sure about, I researched it and learned about it.
As examples, in early January, I spent about ten days reading up on VED (vaccine enhanced disease). Shortly after, I spent a few days digging into chitosan, and trying to understand how sensitive nanoparticle creation is to changes in the mixing process (hint: not very.) Everything I searched for I was able to find, and pretty much everything reinforced the same internally consistent view of the world.
When you find something that doesn't make sense and you're stuck, write it down, file it away and come back to it later. Eventually you'll be able to make sense of it.
When you're able to read through most or all of the whitepaper and understand both what's being discussed and why specific things were selected, you'll be in pretty good shape.
It's not particularly difficult, it just takes time and effort.
↑ comment by johnswentworth · 2021-02-08T17:23:21.476Z · LW(p) · GW(p)
That's a tough question.
To a large extent this project involves specialized knowledge about the immune system, and that's stuff which you just have to look up one way or the other. That part is pretty straightforward. Same with looking up random jargon; that's a part of any biology research.
The harder part is the not-very-legible general intuitions about biological systems - e.g. things like "a <25 amino acid peptide isn't likely to have any function as a protein in its own right" or "error bars are on a log scale, and probably wide". If you want to acquire those sorts of intuitions, one relatively-fast path might be the bionumbers book.
On the other hand, even without those intuitions you could just brute-force your way through by checking everything. For instance, if you don't have the intuition that error bars are wide and on a log scale, you could find a paper experimentally measuring the concentration tolerances for chitosan nanoparticle formation. In some ways that's better - you're less likely to miss things and you can end up a lot more confident in your assessment - but it's a lot more work.
comment by PatrickDFarley · 2021-02-04T18:07:57.241Z · LW(p) · GW(p)
Very impressed by this, I really hope it works. These are the kind of audacious efforts that I love to see in this community
comment by john_a · 2021-02-21T10:17:03.882Z · LW(p) · GW(p)
Quote: “But at this point it’s only ~4 months until I’d get a vaccine anyway, so the price tag is only arguably worthwhile.”
I’d question this assumption, due to the new mutations such as E484K. It’s looking probable that “v1” vaccines will be significantly less effective against it - and v2 vaccines may not be out until end of 2021.
Theoretically, assuming RadVac is effective, once they update for new mutations, you may be able to do a new batch and self-vaccinate quicker than waiting for your turn at v2.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-21T16:58:54.364Z · LW(p) · GW(p)
In addition to this, the current version of radvac was designed to be more robust to mutations on its own. Specifically, it targets some non-spike/RBD parts of the virus, which are more evolutionarily conserved. And of course simply having an immune response to a different part of the virus than the large population of mainstream-vaccinated people is itself helpful, since mutations will be selected to circumvent the most common immune mechanisms.
comment by magfrump · 2021-02-12T06:01:05.814Z · LW(p) · GW(p)
Any preliminary results on side effects so far?
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-12T17:18:23.528Z · LW(p) · GW(p)
Nothing unusual that I've noticed. A bit of congestion a few hours after taking it is expected, but air quality is not great here so I'm mildly congested pretty often anyway.
comment by andrewseanryan (andrewseanryan@gmail.com) · 2021-02-05T20:42:32.428Z · LW(p) · GW(p)
Have you tried making at home rapid covid tests? I have often wondered if it is possible to make an at home version of the spit paper rapid tests. No clue what this would take but would be great since the government hasn’t allowed them yet.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-06T16:42:07.202Z · LW(p) · GW(p)
I'd be very curious to see a protocol for this.
comment by gojomo · 2021-02-05T22:36:31.233Z · LW(p) · GW(p)
Curious, are the peptides-as-ordered shipped in freezer packs?
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-05T22:43:51.718Z · LW(p) · GW(p)
No, they ship dry.
comment by Borasko · 2021-02-04T19:30:11.989Z · LW(p) · GW(p)
Thanks for posting this, I didn't know about radvac before and now I am excited. I will read the white paper and probably make some myself. If your results are good which I really hope they are, I will try to help my family members get some as well. I don't mind paying $1000+ for family safety, and with the delayed vaccine rollout I would feel better even getting the vaccine to them one month earlier. So thanks again! I hope all goes well.
comment by jmh · 2021-02-04T19:29:09.534Z · LW(p) · GW(p)
Thanks for the write up! I started to do this myself but quickly found I was a bit confused on how to even order the peptides. I was expecting to be able to search product lines but.... Not really possible. So...is the process to simply identify some suppliers (not hard to find with Google) give them the amino acid sequences in the instructions and ask for a quote? (Hit that initial speed bump and have not gotten back to looking for phone numbers or customer support chat windows.)
Just a thought. So this doesn't scale well for storage and shipping it sounds but production is pretty simply and materials seem to be fairly durable for storage (I don't think the peptides decay quickly). If so, couldn't the model be that a small lab at the many, many, many pharmacy (or at lest the big chains) stores make batches for the appointments they have scheduled. Given that the delivery method seem to be better (thanks for bringing that up -- was going to ask) and that should take out a lot of the supply chain bottlenecks we have been seeing I would think. (Or once the transition and supporting setup/infrastructure locally is done.)
The other question, having taken this is there any concern about being required later to also take one of the officially blessed vaccines. This might be even more important is you don't show the antibodies for some reason but have reacted to the vaccine. I don't think I completely read the radvac white paper but don't recall them having any section that might have spoken to that.
Again, great write up and contribution.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-04T19:42:41.226Z · LW(p) · GW(p)
So...is the process to simply identify some suppliers (not hard to find with Google) give them the amino acid sequences in the instructions and ask for a quote?
The one I used had an online "custom peptide synthesis" order form. I just pasted in the sequences, checked a few boxes, it autogenerated the price tag on checkout screen, and then I hit the order button. (It actually took a bit longer than that since both my girlfriend and I triple-checked the sequences.) I specifically avoided any suppliers which would have made me talk to a salesperson to get a quote - that crap is a strong sign of Maziness.
having taken this is there any concern about being required later to also take one of the officially blessed vaccines
Dentin had a comment on this somewhere in this rapidly-growing comment section, saying that the commercial vaccines use different epitopes and should therefore play well together with this one.
Replies from: Dentin, ChristianKl↑ comment by Dentin · 2021-02-05T12:13:37.073Z · LW(p) · GW(p)
Yeah, the pfizer vaccine looks like it just uses mRNA to construct the RBD (receptor binding domain) of the spike protein, which is about two hundred amino acids long. None of the default 9 peptides in gen 9 radvac are for that domain. See page 40 of the whitepaper for the full spike protein sequence; the highlighted blue is the RBD, and the short underlined sequences are peptides selected for the vaccine.
The moderna vaccine uses mRNA to construct pretty much the whole spike protein, including the RBD. This has overlap with 3 of the 9 radvac peptides.
This paper has one of the better lists I've found of commercial vaccine types:
https://www.sciencedirect.com/science/article/pii/S2319417020301530
From the list in that paper, it seems like pretty much all commercial products are using spike as the primary target; radvac is unique in that it also targets ORF and Nuc.
-----
Amusingly enough, when talking about the commonly targeted spike protein RBD sequences, the radvac whitepaper lists on page 29: "Spike 450-500; ACE2 binding residues of the RBD (Zhang et al); low degree of conservation; probably moderate to high mutant escape potential". So they pretty much called it in regards to the new virus strains with mutations like E484K.
Another radvac whitepaper quote, which seems to line up with my independent research: "It is important to note that most published neutralizing antibodies target Spike RBD, as do many vaccines in commercial development. However, given the high degree of mutability of the RBD portion of Spike, it is highly recommended to identify and select targets outside the RBD because of mutant escape potential."
And lastly, more specific to why no RBD peptides were selected, page 34:
"Therefore, rather than focusing on ACE2-binding epitopes in the highly mutation prone RBD to inhibit virus binding to the ACE2 receptor, we targeted these B-cell epitopes in the highly conserved portions of the Spike protein to strategically neutralize proteolytic cleavage and membrane fusion. Furthermore, all three are bound by antibodies present in the sera of large fractions of convalescents, and they produce among the highest signals in linear epitope mapping studies, which are far higher than signals measured for binding to any linear epitope in the RBD."
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-05T17:50:08.193Z · LW(p) · GW(p)
Yeah, the pfizer vaccine looks like it just uses mRNA to construct the RBD (receptor binding domain) of the spike protein, which is about two hundred amino acids long.
Holy crap, the entire RBD!? These mRNA vaccines are a technical marvel, I'm amazed they can actually deliver that into a cell safely.
I really hope you write up a post on your learnings, the comments have been amazing and way more in-depth than my knowledge.
↑ comment by ChristianKl · 2021-02-04T23:59:30.829Z · LW(p) · GW(p)
The one I used had an online "custom peptide synthesis" order form.
How about sharing the link?
Dentin had a comment on this somewhere in this rapidly-growing comment section, saying that the commercial vaccines use different epitopes and should therefore play well together with this one.
Besides different epitopes, if the RadVac vaccine gives you an immune reaction in the mucosal immune system and the other vaccines give you a immune reaction in the normal immune system you will want both.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-05T00:12:07.395Z · LW(p) · GW(p)
How about sharing the link?
I don't want people giving the company crap if this post ends up very widely read. If anyone would like a link, PM me.
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-05T00:20:29.399Z · LW(p) · GW(p)
Are we really living in times where "company helped people with doing something themselves against COVID-19" would be problematic PR-wise?
Replies from: mingyuan, Dentin↑ comment by Dentin · 2021-02-05T11:53:36.298Z · LW(p) · GW(p)
Absolutely obviously yes. I have some level of concern that this post will go viral (ha ha), get a lot of attention outside of lesswrong, and the company I'm working with will cancel my order because it's "covid misinformation" related.
The FDA might be slow and take months to approve safe things while thousands of people die per day, but they're perfectly capable of announcing an immediate and indefinite peptide ban in under a day because a news article crossed the wrong person's desk.
Replies from: habryka4↑ comment by habryka (habryka4) · 2021-02-05T21:04:43.692Z · LW(p) · GW(p)
About the "viral" part. This post is currently at the top of HN: https://news.ycombinator.com/item?id=26022750
Replies from: Dentincomment by billmei · 2021-02-12T03:46:32.261Z · LW(p) · GW(p)
Is this blog post potentially a source of X-risk? Popularizing the idea of "mix together chemicals you got in the mail" sounds like an attack vector for an AGI wanting to escape its box.
At the risk of arguing from fictional evidence [LW · GW], Eliezer writes in That Alien Message [LW · GW] of an AGI coming up with a cover story to convince someone outside the box to construct nanomachines (amino acids!) to do its bidding.
We sent messages [...] to labs that did their equivalent of DNA sequencing and protein synthesis. We found some unsuspecting schmuck, and gave it a plausible story [...], and told it to mix together some vials it got in the mail. Protein-equivalents that self-assembled into the first-stage nanomachines, that built the second-stage nanomachines, that built the third-stage nanomachines...
"Make your own COVID-19 vaccine at home!" sounds like a pretty compelling cover story, to the point that nobody on LessWrong has yet commented on the possibility of this being the machinations of a malicious actor, despite this very specific scenario already being wargamed by Eliezer himself!
I'm not accusing the author of secretly being an AI writing this story, I think that's rather unlikely, and a less fictional framing is that maybe normalizing the idea of "it's really easy to try your own biology experiments at home" increases X-risk from accidental or intentional biological hazards created by people who think this blog post is cool and are inspired to try their own experimentation at home.
More generally, if this blog post is an information hazard, what are the norms on LessWrong for discussing or promoting potential attention hazards? Currently we're incentivized to get upvotes for cool, intellectually interesting posts and comments, and we don't get upvotes for keeping information hazards to ourselves. ;)
Replies from: Zack_M_Davis, ChristianKl↑ comment by Zack_M_Davis · 2021-02-12T04:09:16.335Z · LW(p) · GW(p)
I think another John Wentworth post [LW · GW] is applicable here. It's not hard to invent reasons why any given post might increase existential risk by some amount. (What if your comment encourages pro-censorship attitudes that hamper the collective intellectual competence [LW · GW] we need to reduce existential risk?) In order to not function as trolling, you need to present a case for the risk being plausible, not just possible.
Replies from: billmei↑ comment by ChristianKl · 2021-02-12T13:05:17.792Z · LW(p) · GW(p)
Is this blog post potentially a source of X-risk? Popularizing the idea of "mix together chemicals you got in the mail" sounds like an attack vector for an AGI wanting to escape its box.
No, an AGI would already have to escaped it's box to take over companies from which you can order chemicals and sent out what it wants. From there it can simply pay people to do what it wants. EmeraldCloud Lab also exists and could mix out what the AGI wants. Plenty of people take drugs they order on the Darknet.
comment by LThorburn · 2021-02-07T23:30:49.696Z · LW(p) · GW(p)
I ran this by an epidemiologist friend and they pointed out that SARS-CoV-2 is mutating fairly quickly, so wide deployment of low-effectiveness vaccines (possibly including this one) would encourage the emergence and spread of vaccine-resistant strains of the virus.
This might become a big problem because vaccines are being rolled out whilst there is a lot of the virus circulating in the community.
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-10T15:23:13.814Z · LW(p) · GW(p)
That's only an issue if the low effectiveness vaccine mainly targets the spike protein and targets it in ways that the vaccine can mutate out of.
In contrast to the approved vaccines this vaccine is designed to make it harder for the virus to become resistent to it.
Replies from: ka↑ comment by ka · 2021-02-13T20:26:48.622Z · LW(p) · GW(p)
How sure are you that this believe is correct?
Replies from: ChristianKl↑ comment by ChristianKl · 2021-03-05T10:21:43.916Z · LW(p) · GW(p)
There are multiple claims here, if you want to know something specific ask a more specific question.
comment by Douglas_Knight · 2021-02-06T03:41:43.023Z · LW(p) · GW(p)
If it's so cheap and easy to make vaccines, why aren't commercial ones made this way? In particular, the Novavax vaccine sounds similar, so why wasn't that the first vaccine to market?
Added: Specifically, the ultimate purpose of a vaccine is to get protein into the body. Traditional vaccines grow the virus using its own reproductive apparatus. Fancy new vaccines, like the adenovirus and mRNA vaccines inject instructions and induce the subject to manufacture proteins. But if it's so easy to just print proteins, why don't we do that? That's what Novavax does, unlike the ~7 vaccines that beat it to market.
Added: one difference is that all the vaccines that made it to market, including, I think, Novavax, used the whole spike protein, whereas this proposal uses short peptides. Identifying the right short snippets takes time, while using the whole protein is simpler and more likely to work. The cost of peptides is probably super-linear in length. Still, I remain confused about Novavax.
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-12T12:45:18.758Z · LW(p) · GW(p)
Identifying the right short snippets takes time, while using the whole protein is simpler and more likely to work.
If you identify snippets that are conserved over evolutionary time, it's harder for the virus to mutate in a way that it becomes immune to your vaccine.
Specifically, the ultimate purpose of a vaccine is to get protein into the body.
That sense got me to write my last post [LW · GW]. I think there's some hope that bringing the vaccines into cells in the body produces a slightly better immune response then having them in blood and giving an adjuvant.
If it's so cheap and easy to make vaccines, why aren't commercial ones made this way?
That might be the "why does nobody prescribe melatonin"-question that Scott Alexander discussed. You can't patent "we target the spike protein" as that's the obvious thing that everybody is doing.
In particular, the Novavax vaccine sounds similar, so why wasn't that the first vaccine to market?
It seems Novavax spent more time testing different variations before deciding that using the spike protein as everybody else is the way to go.
As far as I understand they also ran their own clinical trials while Moderna and BioNTech got external help running the trials which might made running the trials faster.
The cost of peptides is probably super-linear in length.
This seems to be true for the amount that needs to be produced to do biology at home. If you however optimize hela cells to produce a given protein and then just let them doublicate this gives you proteins that are cheap enough if you do things at scale.
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-12T17:25:49.023Z · LW(p) · GW(p)
If you however optimize hela cells to produce a given protein and then just let them doublicate this gives you proteins that are cheap enough if you do things at scale.
Do people actually do this? I would expect it to be both more expensive and riskier to use HELA cells rather than bacteria, but I've never looked into the details. Do they just not separate the target protein from all the other proteins, and therefore want it mixed with human proteins rather than bacterial proteins?
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-12T21:49:43.747Z · LW(p) · GW(p)
I read to a bunch of different stuff and it might be that it's easier to you some other bacteria here.
comment by kjz · 2021-02-03T22:36:21.535Z · LW(p) · GW(p)
Crazy thought, and I doubt this is likely on large scale or it would have been in the news, but any chance this could explain the higher than expected percentage of nurses who have rejected getting the vaccine? Perhaps some have already vaccinated themselves under the radar! And therefore have no need to take the "real" one.
Replies from: jayterwahl, Dentin, cistran, ChristianKl, eigil-rischel↑ comment by jayterwahl · 2021-02-04T10:23:45.904Z · LW(p) · GW(p)
I predict that is an overly-optimistic reason for why they're rejecting the vaccine.
↑ comment by Dentin · 2021-02-04T12:18:16.224Z · LW(p) · GW(p)
My personal estimate is that the the percentage of nurses who have done this is effectively zero (less than one in a thousand with high probability, less than one in ten thousand with moderate probability.)
Further, those who did do it are likely to have read through the whitepaper, and therefore are also likely to get the commercial vaccine, as it covers different epitopes than the radvac vaccine.
Replies from: kjz↑ comment by kjz · 2021-02-04T14:53:44.707Z · LW(p) · GW(p)
Agree it is extremely unlikely that many nurses have done so, and your probabilities seem quite reasonable. I think the main reason why many nurses have declined the vaccine is social signaling - either to maintain their social status within a mostly anti-vaccine peer group, or to maintain credibility with their anti-vaccine patients, who may be reluctant or outright refuse to be treated by a nurse who has been vaccinated because such a nurse is on "the wrong side" and can no longer be trusted. However, a nurse could self-administer the radvac vaccine and get some protection, while still being able to honestly claim they have no plans to get the commercial vaccines.
I hadn't read the whitepaper yet before my initial post, and after a quick scan it looks like you are correct that radvac covers different epitopes than the commercial vaccines (I haven't done my own detailed analysis yet). Are you and others planning to take radvac still planning to get a commercial vaccine once you are eligible?
Replies from: Dentin↑ comment by Dentin · 2021-02-04T22:50:03.394Z · LW(p) · GW(p)
Yes, I still plan to get the commercial vaccine once it's available to me (likely some time in august.) As I understand it, the commercial vaccines hit different areas of the virus from the ones that radvac selected, improving protection even further.
There is actually an optional peptide for radvac which does cover one of the same regions as the commercial vaccines. I elected not to include it under the assumption I'd be getting it from the commercial vaccine.
↑ comment by cistran · 2021-02-09T09:59:19.795Z · LW(p) · GW(p)
This is a conversation with an MD I recorded which may answer some of the reluctance:
tldr; An untested delivery mode scares an MD to think that neurodegenerative diseases will increase in vaccinated.
I think lipid nanoparticles may have too broad a tropism, far broader even than attenuated virus vaccines (which are still limited to the tropism of the wild-type virus), and thus could pose a uniquely high safety hazard due to cytotoxic attack on the broad cellular range that uptakes the LNPs. Since the LNPs would enter cells via endocytosis, the SARS-CoV-2 epitopes would be expressed on MHC-1 molecules, making them targets of cytotoxic CD8 lymphocytes, attacking a much greater range of cells than any previous vaccine modality. This is concerning in general, but it’s a nightmare scenario if the vaccines are crossing the blood-brain barrier and endocytosing into e.g. oligodendrocytes (multiple sclerosis risk) or motor neurons (which could possibly cause an ALS-type picture). No other vaccine has this broad tropism. That is THE major safety concern here.
1. Because of the anatomy and circulatory trajectory from the deltoid and cephalic vein (essentially a straight shot into the SVC), if enough “spillover LNPs” are getting shuttled into the right atrium and transiting through the pulmonary circulation — which could be high, another reference here for the rich vasculature around IM injection — then one of their earliest stops on the map after exiting the heart would be in tissues serviced by branches of the common carotid and subclavian arteries (including the CNS), enhancing delivery to tissues behind the blood-brain barrier simply due to higher relative concentration at tissue corridors more proximal to the injection site.
2. Even if initial transit through the BBB and into other sensitive tissue parenchyma is relatively low, there’d be a cumulative effect with each booster delivering more spillover LNPs to those non-local sites.
3. Related to that point, the duration of immunity is still unclear, and there seems to be general agreement that while COVID-19 symptoms are reduced with the immunization, viral spread is not. If antibody and memory B/T-cell levels wane within a few months after vaccination, then we’d be looking at repeated boosters possibly multiple times a year given ongoing community dissemination. And since the development of many e.g. CNS disorders is gradual — with subclinical issues taking shape over years before clinical manifestations become apparent (as seen in MS and ALS) — such cumulative damage likely wouldn’t raise red flags at first, but could increase in likelihood with successive boosters.
↑ comment by ChristianKl · 2021-02-04T17:07:00.558Z · LW(p) · GW(p)
Given that this vaccine targets a different part of the immune system there's no good reason to reject an injection vaccine when you take this vaccine.
Replies from: kjz↑ comment by kjz · 2021-02-04T18:30:27.545Z · LW(p) · GW(p)
For the average Less Wrong reader, I tend to agree. But a nurse in an area with a strong, vocal anti-vaccine community may face substantial social pressure to (at least publicly) reject commercial vaccines, for the reasons I stated above.
Replies from: ChristianKl↑ comment by ChristianKl · 2021-02-05T00:29:12.046Z · LW(p) · GW(p)
The average nurse in a anti-vaccine community is not going to make their own vaccines. I would also expect that most nurses will face some social pressure in their workplace to take the vaccine.
↑ comment by Eigil Rischel (eigil-rischel) · 2021-02-04T10:24:35.959Z · LW(p) · GW(p)
This seems prima facie unlikely. If you're not worried about the risk of side effects from the "real" vaccine, why not just take it, too (since the efficacy of the homemade vaccine is far from certain)?. On the other hand, if you're the sort of person who worries about the side effects of a vaccine that's been through clinical trials, you're probably not the type to brew something up in your kitchen based on a recipe that you got off the internet and snort it.
comment by CharlieMarr@protonmail.com · 2021-02-26T11:32:45.763Z · LW(p) · GW(p)
This seems like a cool and interesting experiment, and it seems rational from a "zoomed-in" perspective. i.e. "if I'm going to get vaccinated, I might as well do it myself, nasally, with fewer ingredients....the potential benefits are greater than the costs. Especially when considering adjacent benefits like learning, being a trailblazer, potentially advancing knowledge, etc."
But if you "zoom out" and take a broader view of the whole Covid phenomenon, it seems like you may be (irrationally?) accepting a number of big assumptions before beginning your cost-benefit analysis. It seems that you may be assuming some subset of the points below:
a) You face significant risks from Covid-19 even though you are not elderly and don't have comorbidities
b) A vaccine is an important part of reducing your risk of severe Covid-19 outcomes. (You don't think you can achieve sufficient risk reduction through preventative risk reducers like Vitamin D, sleep, exercise, stress reduction, etc., and you don't think you can achieve sufficient risk reduction through therapeutics like steroids, antivirals, antiparasiticals, etc.)
c) Vaccine experts are generally reliable sources of data and insight. (Or at least you can successfully navigate the challenge for distinguishing real experts from fake, and you are fully aware of the conflicts faced by researchers and public health experts due to their funding sources, political pressures, regulatory capture by industry, etc.)
d) The scientific (or bureaucratic?) challenges that prevented any previous coronavirus vaccines from being approved for humans were solved in the past 10 months, not by haste, but by scientific breakthroughs or by the realization that previous non-approval was in error.
e) To the extent that proof of vaccination or antibodies are required for things like travel, etc., your DIY vaccine and/or resulting antibodies will be accepted by authorities. You would not be required to get, say, the Pfizer or Moderna vaccines on TOP of your DIY vaccine. Or, if you were, doubling up on complementary vaccines would be neutral or positive, not negative.
Anyway, I'd be curious to hear any of your thoughts on these assumptions. I loved your post, and I love the DIY decision at a micro-level. If you want a vaccine, why NOT a quicker DIY nasal one instead of waiting. But I find myself wondering about what principles, assumptions, and decisions you embraced in advance of the DIY vs waiting for Pfizer decision. Thanks for any insight you can share!
Replies from: johnswentworth↑ comment by johnswentworth · 2021-02-26T18:43:47.371Z · LW(p) · GW(p)
At this point in the game, things like "advancing knowledge" and "I really want to know if it was actually this easy all along" are at least as big a factor as the object-level benefits. If this were last July, or if I lived in Europe, then it might be a different story, but at this point I'm likely to get vaccinated within a few months anyway.
On your specific points:
a) COVID still sucks, I don't want my food to taste like nothing for six months, etc, but I'm definitely not at any significant risk of death from COVID.
b) I definitely do not expect that sleep/exercise/vitamin D/etc would achieve anywhere near the degree of risk reduction that a vaccine provides, and I do expect preventative to be way better than treatment here.
c) I certainly would not consider "vaccine experts" reliable, as a category. I do think that I am extremely unusually good at distinguishing real experts from fake. Indeed, there's an argument to be made that this is the major thing which we should expect rationalists to be unusually good at. In the case of RadVac specifically, I think it is considerably more likely to work than a typical vaccine trial, and typical vaccine trials have ~40-50% success rates to start with (the highest success rate of any clinical trial category).
d) Not sure what you mean by this one. Sounds like you're making a generalized efficient markets argument, but I'm not sure exactly what the argument is.
e) Antibody test results would be the goal here. Though in practice, most governments seem so incompetent that they're not even actually looking at antibody test results or vaccination, so it's not a very large factor.
Replies from: CharlieMarr@protonmail.com↑ comment by CharlieMarr@protonmail.com · 2021-03-01T20:31:16.441Z · LW(p) · GW(p)
Thank you so much for your great answers.
I have one follow-up question for you.
I really liked/enjoyed your logic on why you decided to make your own vaccine. You seem like a highly rational guy, and because of that, I find myself curious about your logic on the preceding decision--the decision that you would take a brand-new Covid-19 vaccine, and soon, from, say, Pfizer.
Most people are eager to get that vaccine, but most people are not rationalists. I'm curious about the logic an intelligent and skeptical rationalist used to decide he'd take a new Pfizer vaccine.
I'm curious about how you came to the conclusion that a new vaccine would be way better than general good health and treatment options. And I'm curious about how you came to the conclusion that a new, experimental, lightly tested, approved-only-under-emergency-authorization vaccine from a politically powerful, profit-seeking corporation poses fewer risks to you than a case of Covid-19.
Most people are enthusiastic about the new vaccines. But the enthusiasm in many cases is driven more by a year of wall-to-wall media coverage than by any logical consistency. Many of the millions rushing to get Covid-19 vaccines are not up to date on the CDC's adult vaccination schedule and do not get flu shots.
Most people don't even try to distinguish real experts from fake. When it comes to Covid-19, most people unquestioningly accept scientific pronouncements from...politicians and journalists. (And politicians and journalists, however noble their intentions, sometimes can't help but be influenced by the the pharmaceutical industry's massive lobbying efforts and massive advertising spend in the news media.)
You, on the other hand, do try to distinguish real experts from fake, and you say you are extremely unusually good at it. And as a rationalist you're probably much more likely than the average person to understand incentives, follow the money, consult primary scientific sources, and more accurately estimate risks and rewards.
And that's why I'm curious about your thought process. Even if your thought process was fairly quick, leveraging previous knowledge. I've seen plenty of emotional or political or conformist reasons for rushing out to get the Pfizer Covid-19 vaccine. But I'm interested in the rationalist reasons for doing it.
(Thanks for reading this reply. I know it's a little long, a little off topic, and tinged with a little cynicism. But I'd love to read any thoughts you care to share about how you made the overall yes/no vaccine decision that preceded your decision to make your own!)
Replies from: johnswentworth↑ comment by johnswentworth · 2021-03-25T23:34:38.779Z · LW(p) · GW(p)
I've been putting off answering this, because a proper answer would require diving into a lot of disparate evidence for some background models. But developments over the past few weeks have provided more direct data on the key assumption, so I can now point directly to that evidence rather than going through all the different pieces of weaker prior information.
The key assumption underlying my belief (and, presumably, most other peoples' belief) in the efficacy of the commercial vaccines is that the data from the clinical trials is basically true and representative. If we believe the data, then the health risks are trivial (compared to COVID), and the effectiveness is decent, and our certainty about these two facts is very high. The vaccines are not "lightly tested" in any epistemically-relevant sense, only relative to the frankly-unnecessary extreme over-testing typically used. (Zvi's posts [? · GW] frequently provide helpful snapshots of the relevant data/analysis.)
The question which you seem to be interested in is whether the data is trustworthy. The key model here is that, if there were anything even remotely suspicious in there, then regulators and the media would absolutely freak out over it. That's exactly what we've seen over the past few weeks - first with the AZ clotting thing, then with AZ calling their vaccine "79% effective" when the regulators thought it only merited "69 to 74% effective". The clotting thing in particular is a clear case where there was absolutely no real, important problem, and regulators/media freaked out over it anyway, because they are super-over-sensitive to even the tiniest hint of a problem. That follows directly from their incentives: regulators do not get punished for delaying good vaccines, but if a bad vaccine gets regulatory approval, then there's a scandal and an Official Investigation and "heads will roll".
comment by ChristianKl · 2021-02-05T17:11:21.911Z · LW(p) · GW(p)
If nasal application doesn't provide a blood immune response it might be worth testing whether anal application will lead to a blood immune response.
comment by IlyaShpitser · 2021-02-09T19:40:16.318Z · LW(p) · GW(p)
Recommending this to others seems to be coming pretty close to health fraud.
The reasonably ponderous systems in place for checking if things work and aren't too risky are there for a reason.
↑ comment by habryka (habryka4) · 2021-02-10T21:01:36.819Z · LW(p) · GW(p)
Please name those reasons instead of vaguely alluding to them. The above post and many other writings on LessWrong address some of the most commonly made arguments for why there is indeed not a particularly good reason for the incredibly slow process of the FDA trial system. I would be glad to hear more arguments, but am not very excited about vague allusions without any substance.